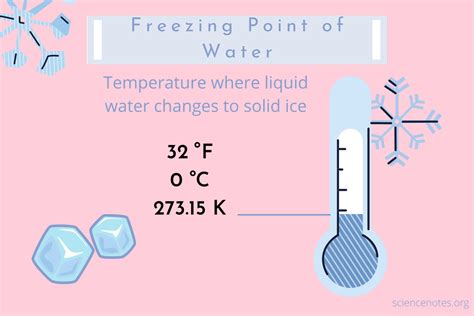
Understanding the freezing point of water is crucial for both scientific inquiry and everyday practicality. Water, a fundamental substance, freezes at 0 degrees Celsius (32 degrees Fahrenheit) under standard atmospheric pressure. This deceptively simple fact has profound implications for fields ranging from cryogenics to meteorology, and it holds a straightforward but critical role in daily life scenarios like freezing and thawing processes in cooking and climate impact studies.
Key Insights
- Water freezes at 0 degrees Celsius (32 degrees Fahrenheit), under standard atmospheric pressure.
- Pressure variations can slightly alter the freezing point.
- Understanding the freezing point of water is vital for applications in science, cooking, and climate studies.
Water’s freezing point is foundational to many scientific principles. For instance, cryogenics — the study of extremely low temperatures — relies heavily on this basic principle. Specialized equipment and techniques are employed to reach and maintain temperatures below the freezing point, facilitating the preservation of biological samples and the study of unique material states. One practical application is in the storage of vaccines where precise temperature control is critical to maintaining efficacy.
Another area where understanding the freezing point of water is invaluable is in meteorology. Weather patterns and climate studies depend on knowing how water behaves under various temperature conditions. For example, understanding how water freezes can provide insights into phenomena such as frost formation, which impacts agriculture and infrastructure. By monitoring and predicting these processes, meteorologists can issue timely warnings, preventing potential damage from freezing weather events.
Beyond science and weather, the knowledge of water’s freezing point is essential in everyday cooking and food preservation. For instance, knowing that water freezes at 0 degrees Celsius allows cooks to effectively freeze ingredients, which is a crucial step in making ice cream or preserving fresh produce for later use. Additionally, when dealing with issues like thawing frozen food items, understanding the freezing point ensures safety and quality by avoiding the refreezing of thawed items without proper handling.
Does pressure affect the freezing point of water?
Yes, slight variations in pressure can affect the freezing point of water. Generally, increasing pressure raises the freezing point, while decreasing pressure lowers it. For example, water can freeze at slightly higher temperatures under higher pressures, which is why the pressure in deep ocean environments can keep water from freezing.
Why is it important to know the freezing point of water for scientific experiments?
Knowing the freezing point of water is crucial for scientific experiments because it allows researchers to maintain the necessary conditions for various experiments. Whether it's studying the effects of cold on biological samples or ensuring the right environmental controls for experiments in chemistry and physics, precise temperature regulation hinges on understanding water's freezing point.
This article underscores the universal importance of knowing the freezing point of water. From scientific research to daily cooking, this fundamental property of water enables precise control and understanding across various fields. It is a simple yet profound truth that underpins much of our scientific knowledge and everyday practices. Understanding this fundamental concept ensures better outcomes in diverse applications, from preserving life-saving vaccines to safeguarding our food supply.