P Delta G in chemistry is a crucial concept that provides insight into the spontaneity and feasibility of chemical reactions. The term “Delta G” refers to the change in Gibbs free energy, which integrates enthalpy and entropy changes to predict the likelihood of a reaction occurring without input of external energy. Understanding this concept not only deepens our comprehension of chemical thermodynamics but also enhances our ability to design and optimize chemical processes.
Key Insights
- Primary insight with practical relevance: Delta G helps determine the spontaneity and feasibility of a chemical reaction.
- Technical consideration with clear application: A negative Delta G indicates a spontaneous reaction, while a positive Delta G suggests a non-spontaneous one.
- Actionable recommendation: Utilize Delta G calculations to predict reaction outcomes and optimize industrial processes.
The Role of Gibbs Free Energy in Chemical Reactions
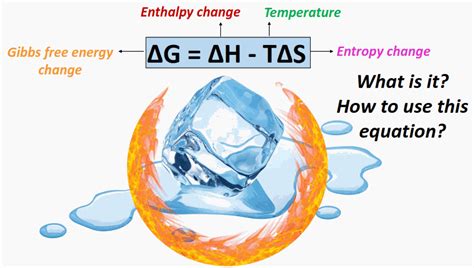
In chemical thermodynamics, Gibbs free energy (G) combines enthalpy (H) and entropy (S) changes to quantify the energy available to do work under constant pressure and temperature conditions. The formula to determine the change in Gibbs free energy (ΔG) is:ΔG = ΔH - TΔS
Here, ΔH represents the change in enthalpy, T is the absolute temperature in Kelvin, and ΔS stands for the change in entropy. This equation succinctly captures the essence of how energy transformations drive chemical reactions.
The Significance of Delta G in Spontaneity
The sign of Delta G is paramount in determining the spontaneity of a reaction.If ΔG < 0, the reaction proceeds spontaneously, meaning the system can evolve into a state of lower free energy without requiring additional external energy input. Conversely,
if ΔG > 0, the reaction is non-spontaneous, necessitating an external energy source to proceed. The closer ΔG is to zero, the closer the system is to equilibrium, indicating that the reaction is almost at equilibrium.
For example, consider the reaction between hydrogen gas and oxygen gas to form water:
2H₂(g) + O₂(g) → 2H₂O(l)
This reaction has a very negative ΔG, indicating it is highly spontaneous. In industrial settings, such knowledge allows for the design of efficient and cost-effective processes for producing water and other chemicals by leveraging reactions with favorable ΔG values.
What is the relationship between Delta G and reaction equilibrium?
At equilibrium, the Gibbs free energy change (ΔG) is zero. This means that the system's free energy is minimized, and no net change occurs. Thus, any attempt to push the reaction in either direction will be met with an equal and opposite force, maintaining equilibrium.
Can Delta G change over the course of a reaction?
Yes, Delta G changes over the course of a reaction as reactants are converted into products. Initially, ΔG can be positive, negative, or zero, depending on the stage of the reaction. However, as the reaction progresses toward equilibrium, ΔG will decrease to zero, indicating the system has reached a state of equilibrium.
In conclusion, Delta G is an essential parameter in chemistry that encapsulates the energetics of chemical reactions. By calculating ΔG, chemists can predict whether a reaction is spontaneous, design efficient chemical processes, and ensure that reactions proceed as intended. This understanding not only advances scientific knowledge but also drives practical applications across various industrial and technological fields.