Welcome to this comprehensive guide on the wave mechanical model, a fundamental concept in quantum mechanics that explains the behavior of particles in terms of wave-like properties. The wave mechanical model brings clarity to many quantum phenomena, from the double-slit experiment to atomic orbitals. Whether you're a student, an engineer, or simply a curious mind, this guide will demystify the wave mechanical model and offer you actionable insights to apply in practical scenarios.
Understanding the Wave Mechanical Model: A Problem-Solution Approach
The wave mechanical model, or wave mechanics, offers a profound way to understand the behavior of particles at atomic and subatomic levels. Many traditional concepts in physics, like the deterministic paths of classical mechanics, give way to probabilistic explanations that make sense only in quantum mechanics. This shift often poses challenges for those new to the field. How can we, as users, navigate this complex terrain and extract meaningful information? Here’s where our guide comes into play. It not only elucidates the theoretical underpinnings of wave mechanics but also bridges the gap between theory and practical application, ensuring you get tangible benefits from your understanding of quantum mechanics.
By the end of this guide, you will be equipped with the knowledge to apply wave mechanics principles in problem-solving, from analyzing atomic structures to understanding quantum computing. Let’s dive in and unravel the mysteries of quantum mechanics with confidence.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start visualizing particles as waves using Schrödinger’s equation to understand their behaviors.
- Essential tip with step-by-step guidance: Use the particle-in-a-box model to predict electron probability distributions in atoms.
- Common mistake to avoid with solution: Avoid thinking of particles as rigid bodies; embrace the wave-like nature instead to avoid misunderstandings.
Exploring Schrödinger’s Equation: The Core of Wave Mechanics
At the heart of the wave mechanical model lies Schrödinger’s equation, which governs how the quantum state of a physical system changes over time. This equation is pivotal in quantum mechanics because it provides a mathematical description of the wave function—a complex number function representing the probabilities of a quantum system.
Let’s break down the essentials:
- The Schrödinger equation comes in two forms: the time-dependent form and the time-independent form.
- The time-dependent Schrödinger equation is given by:
- Form: iħ∂ψ/∂t = Hψ
- Where:
- i is the imaginary unit.
- ħ (h-bar) is the reduced Planck’s constant.
- ∂ψ/∂t is the partial derivative of the wave function ψ with respect to time t.
- H is the Hamiltonian operator.
- ψ is the wave function.
The time-independent Schrödinger equation is essential for stationary states:
- The time-independent form is given by:
- Form: Hψ = Eψ
- Where:
- H is the Hamiltonian operator.
- ψ is the wave function.
- E is the energy eigenvalue associated with the wave function.
To understand the practical application:
- Start by identifying the potential energy V(x) for your system.
- Set up the Hamiltonian operator H, which typically consists of the kinetic energy operator T and potential energy V(x):
- Form: H = T + V(x)
- Apply the Schrödinger equation to solve for the wave function ψ by using techniques such as separation of variables or numerical methods.
- Once you have the wave function, compute expectation values for physical quantities such as position, momentum, and energy.
Visualizing Particles as Waves: Practical Examples
Visualizing particles as waves helps bridge the abstract concept of wave mechanics with real-world applications. Here are practical examples to illustrate how wave mechanics applies:
- The Double-Slit Experiment:
In this classic experiment, particles such as electrons are fired at a barrier with two slits. Instead of showing individual trajectories, the particles create an interference pattern on a detection screen, indicative of wave-like behavior.
Understanding this experiment’s outcome using wave mechanics means recognizing that each particle acts as a wave that interferes with itself, creating constructive and destructive interference patterns.
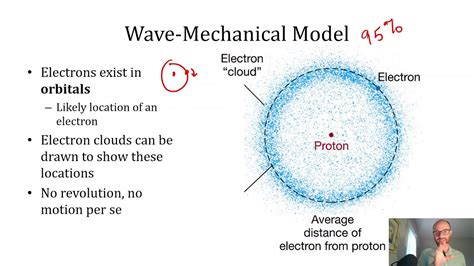
- Atomic Orbitals:
Electrons in an atom exist in probabilistic regions called orbitals, described by the wave function. By solving the Schrödinger equation for the hydrogen atom, we obtain the well-known shapes of s, p, d, and f orbitals.
These shapes provide insight into where electrons are most likely to be found, which is crucial for understanding chemical bonding and atomic spectra.
- Quantum Tunneling:
This phenomenon occurs when particles penetrate through potential barriers, something classically impossible but explained by wave mechanics. Quantum tunneling is fundamental in semiconductor devices and nuclear fusion.
FAQs on Wave Mechanical Model
Why is Schrödinger’s equation so important?
Schrödinger’s equation is the cornerstone of wave mechanics because it provides a mathematical framework to determine the wave function of a quantum system, which in turn allows us to predict probabilities of finding a particle in various states. Understanding this equation is fundamental to grasping many quantum phenomena.
Can I use Schrödinger’s equation for any quantum system?
While Schrödinger’s equation provides a powerful tool, it is most commonly applied to single, non-relativistic particles. For complex systems involving many particles or relativistic effects, additional modifications and extensions of the equation are necessary. Nevertheless, the core principles remain valid across various applications.
What are some practical applications of wave mechanics?
Wave mechanics has diverse practical applications, ranging from explaining chemical bonding and atomic spectra to facilitating the development of quantum computing and semiconductor technology. Understanding wave mechanics also enhances our comprehension of nuclear processes and even the behavior of electrons in mesoscopic systems.
We hope this guide has illuminated the intricacies of the wave mechanical model and equipped you with the necessary tools to apply quantum mechanics in practical contexts. Embrace the wave-like nature of particles, solve Schrödinger’s equation for different systems, and always keep in mind the probabilistic nature of quantum phenomena. Happy exploring!