Understanding the Master Van der Waals Formula through Simplified Perspectives
In the realm of thermodynamics and physical chemistry, the Van der Waals formula serves as a pivotal bridge connecting gas laws to molecular interactions. While its formal derivation and complex algebraic structure might initially seem daunting, demystifying it through simplified interpretations and practical insights can unveil its underlying elegance. This article offers a thorough yet accessible examination of the Van der Waals formula, aiming to illuminate its significance for professionals and students alike.
Key Insights
Key Insights
- The Van der Waals formula corrects the ideal gas law by considering real gas molecular interactions.
- It incorporates two crucial parameters: the attraction and repulsion between gas molecules, allowing for more accurate predictions.
- A practical recommendation: Use the Van der Waals equation for real-world gas behavior rather than relying solely on the ideal gas law.
Exploring the Essence of the Van der Waals Equation
The traditional ideal gas law, expressed as PV=nRT, assumes that gas molecules neither attract nor repel each other and have negligible volume. However, in reality, gas molecules exhibit intermolecular forces and occupy a small volume compared to the container they inhabit. To address these discrepancies, the Van der Waals formula refines the ideal gas law by introducing two correction terms: one for the attraction between molecules (a) and another for the volume occupied by the molecules (b).
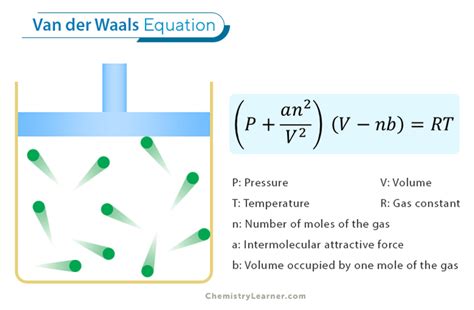
The modified equation is presented as (P + (an^2/V^2))(V-nb) = nRT. Here, ‘a’ adjusts for the attractive forces that pull molecules closer together, reducing the gas pressure. Conversely, ‘b’ compensates for the finite volume of gas molecules, ensuring a more accurate volume reading. By incorporating these factors, the Van der Waals formula provides a more precise model for real gases.
Evaluating the Van der Waals Correction Terms
At the heart of the Van der Waals formula are its correction parameters ‘a’ and ‘b’, each representing different types of molecular interactions. The ‘a’ term is a measure of the attractive van der Waals forces between gas molecules. These forces play a crucial role in lowering the pressure of a real gas compared to what the ideal gas law predicts. When molecules come closer, the attractive forces counteract the kinetic energy of the molecules, leading to a decrease in pressure.
The ‘b’ term quantifies the volume occupied by the gas molecules themselves, adjusting the volume calculation in the formula to reflect this non-zero molecular volume. For example, nitrogen gas (N2) under normal conditions has molecules with a negligible individual volume when compared to the overall volume of the gas. However, as the pressure increases or the temperature decreases, these volumes can become significant, affecting gas behavior markedly.
Real-World Application of the Van der Waals Formula
The practical application of the Van der Waals formula is extensive. It’s particularly useful in fields requiring precise gas behavior analysis, such as chemical engineering, meteorology, and materials science. For instance, when designing compressed gas storage systems, engineers use this formula to predict the behavior of gases under varying temperatures and pressures. Moreover, understanding the Van der Waals interactions is crucial in studying liquid-gas equilibria and designing advanced materials where molecular interactions are a key design parameter.
FAQ Section
Why isn't the ideal gas law sufficient?
The ideal gas law assumes that gas molecules do not interact with each other and occupy no volume, which is not true at high pressures and low temperatures. The Van der Waals formula corrects for these limitations, providing a more accurate representation of gas behavior.
How do 'a' and 'b' parameters differ between gases?
The 'a' and 'b' parameters vary significantly depending on the type of gas due to differences in molecular size and intermolecular forces. For instance, gases like carbon dioxide (CO2) have higher 'a' values due to stronger intermolecular attractions compared to gases like helium (He), which have much lower 'a' values due to weaker intermolecular forces.
With a robust understanding of the Van der Waals formula, one can appreciate its role in bridging the gap between theoretical gas laws and real-world applications, offering a nuanced view of the complexities of gas behavior.