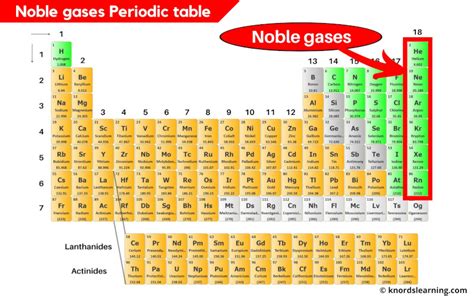
The essential table of elements, specifically focusing on the noble gases, is a subject of immense importance within the field of chemistry. Understanding noble gases is not just an academic exercise; it carries practical implications for a variety of industries including medicine, aerospace, and materials science. Noble gases such as helium, neon, argon, krypton, xenon, and radon are known for their chemical inertness due to having a complete outer electron shell, making them valuable in various technological and scientific applications.
Key insights
Key Insights
- Chemical inertness: Noble gases are highly stable and rarely react under standard conditions.
- Industrial applications: Noble gases have specific uses in lighting, welding, and medical imaging.
- Actionable recommendation: Invest in understanding the unique properties of noble gases for innovation in technology and industry.
Noble gases play a critical role in a variety of applications due to their unique physical and chemical properties. One of the primary reasons for the practical significance of noble gases lies in their chemical inertness. Helium, the second lightest and the second most abundant element in the observable universe, finds its applications in cryogenics, particularly in cooling superconducting magnets in MRI machines and in the helium-cooled centrifugal compressors in gas turbines. Its low boiling point and minimal reactivity make it an ideal cooling agent in extreme conditions.
Another aspect worth examining is the use of neon in lighting. Neon gas is well-known for its use in neon signs and lighting. When electrified, neon emits a vivid red-orange light, a phenomenon that can be explained by the excitation of its electron states. This property has made neon invaluable in the advertising industry, providing vibrant and eye-catching displays that capture attention across busy streets and urban landscapes.
Argon, on the other hand, is widely used in welding due to its ability to shield the weld area from the oxygen in the air, which can cause impurities that weaken the metal. This makes argon essential in various welding techniques like TIG (Tungsten Inert Gas) welding, ensuring high-quality and precise welds in a variety of materials.
Unique Characteristics of Noble Gases
Noble gases possess a range of unique characteristics that distinguish them from other elements. Their full valence electron shells mean they have little tendency to gain or lose electrons, resulting in their well-known inertness. This chemical stability is what grants them their exceptional properties and diverse applications. For example, krypton and xenon can be chemically activated under specific conditions to form compounds such as xenon hexafluoride (XeF6), although these are rare and typically unstable.Xenon is also used in medical applications, particularly in anesthesia. Xenon gas has anesthetic properties similar to those of nitrous oxide (laughing gas) but with a much faster onset and offset of action. Its ability to act as a general anesthetic makes it a valuable resource in medical settings, providing a safer alternative to traditional anesthetics.
Environmental and Future Implications
The environmental impact of noble gases is generally minimal due to their inert nature; however, their extraction and industrial use can have notable ecological effects. For instance, the mining of argon often involves the extraction of large volumes of natural gas, which can lead to environmental concerns related to methane emissions. Furthermore, radon, a naturally occurring noble gas, is a byproduct of the decay of uranium and can be found in soil and water, raising public health concerns due to its radioactivity.Looking ahead, research into noble gases continues to uncover new possibilities. Advances in technology may lead to novel applications, such as using noble gases in nanotechnology or developing new compounds that could revolutionize chemical synthesis.
FAQ section
Can noble gases form compounds?
Under specific conditions, noble gases can form compounds. Although rare, examples include xenon hexafluoride (XeF6) and krypton fluoride (KrF2). These compounds demonstrate the ability of noble gases to form bonds under certain circumstances.
What is the primary use of neon gas?
Neon gas is primarily used in lighting, particularly for neon signs and lighting applications due to its ability to emit a vivid red-orange light when electrically charged.
With this essential table of elements noble gases guide, we’ve examined their unique inertness, diverse applications, and future potential, making it clear why noble gases remain a topic of both scientific and industrial interest.