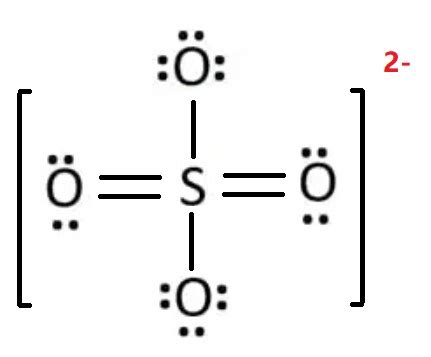
Understanding the sulfate Lewis structure is a crucial step in mastering the fundamentals of chemistry, especially for those who are delving into molecular bonding and geometry. This guide will walk you through the steps to accurately draw the sulfate ion (SO₄²⁻) Lewis structure while addressing common problems faced by students and providing actionable solutions to them.
If you’re struggling with visualizing how to create a clear and accurate Lewis structure for sulfate, this guide is for you. Here, we'll break down the process into manageable steps, provide real-world examples, and offer practical advice to help you avoid typical mistakes. By the end of this guide, you’ll have a solid grasp of the sulfate ion’s structure and how it applies to broader chemical principles.
Getting Started with Lewis Structures
Lewis structures are a visual representation of the valence electrons in atoms and molecules. Understanding how to construct these structures is foundational for comprehending molecular geometry and chemical bonding. The sulfate ion, despite its complexity, follows a set pattern when you break it down into simple steps.
Problem-Solution Opening
Many students find drawing the sulfate ion's Lewis structure challenging because it involves balancing multiple oxygen atoms around a central sulfur atom and correctly distributing the electron pairs. The confusion often lies in determining how many lone pairs to include and how to arrange the atoms to satisfy the octet rule for all atoms involved. This guide will provide you with clear, step-by-step guidance and real-world examples to ensure you can confidently draw the sulfate ion’s Lewis structure.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by identifying all valence electrons from sulfur and oxygen atoms, which will help you outline the initial electron distribution.
- Essential tip with step-by-step guidance: Follow the step-by-step method for placing atoms and electron pairs to avoid common errors like miscounting valence electrons.
- Common mistake to avoid with solution: Don’t forget to form double bonds between the sulfur atom and two of the oxygen atoms. Mistaking this can lead to an incorrect Lewis structure.
Detailed How-To Sections
Step-by-Step Guide to Drawing the SO₄²⁻ Lewis Structure
Drawing the Lewis structure for sulfate involves a series of clear and logical steps. Here’s how to do it:
- Determine the Total Number of Valence Electrons:
First, calculate the total number of valence electrons. Sulfur has 6 valence electrons, and each of the four oxygen atoms contributes 6 valence electrons. Considering the sulfate ion carries a -2 charge, you add two additional electrons. Therefore, the total is:
- 6 (from sulfur) + 4 x 6 (from oxygen) + 2 (for the -2 charge) = 32 valence electrons
- Place the Least Electronegative Atom in the Center:
In the sulfate ion, sulfur is the central atom. Place the sulfur atom in the center and surround it with the four oxygen atoms.
- Arrange Electron Pairs and Form Bonds:
Place single bonds between the sulfur atom and each of the four oxygen atoms. This accounts for 8 electrons (2 for each bond). With 24 electrons remaining, start placing them as lone pairs on the surrounding oxygen atoms.
- Complete the Octet Rule for Outer Atoms:
Each oxygen atom requires 6 more electrons to complete its octet (to make a total of 8). Start placing lone pairs to satisfy these requirements while ensuring that each oxygen atom gets two lone pairs. This accounts for 16 electrons.
- Complete the Octet Rule for the Central Atom:
Sulfur initially has only 4 electrons from the single bonds. To satisfy its octet, sulfur will form double bonds with two of the oxygen atoms. This requires sharing two additional pairs of electrons, accounting for 4 more electrons.
- Check Your Structure:
Verify that each atom follows the octet rule and the total number of electrons matches the calculated total. The structure should now have sulfur at the center with double bonds to two oxygens and single bonds to the other two oxygens, all surrounded by lone pairs on the oxygen atoms to complete their octets.
Practical FAQ
Why is it important to form double bonds between sulfur and two of the oxygens?
Forming double bonds between sulfur and two of the oxygens ensures that sulfur uses only 4 of its valence electrons (compared to 8 if it only had single bonds). This distribution allows the remaining valence electrons to be distributed as lone pairs on the oxygens, which helps satisfy the octet rule for all atoms involved.
By following these steps, you’ll find it much easier to draw the Lewis structure of the sulfate ion and understand the underlying principles of electron sharing and molecular bonding.
Common Pitfalls and How to Avoid Them
When drawing the sulfate ion’s Lewis structure, several common pitfalls can arise:
- Miscounting Valence Electrons: Ensure that you correctly count all valence electrons, including those from sulfur, oxygen, and the ionic charge. This is crucial for maintaining accuracy throughout the structure drawing process.
- Incorrect Bond Placement: Remember that sulfur forms double bonds with two oxygens. Mistaking this can lead to an incorrect distribution of electrons, so always place double bonds carefully.
- Overlooking Lone Pairs: Don’t forget to place lone pairs on the oxygens. If you miss even one pair, it will throw off the entire structure, as it will fail to satisfy the octet rule.
By keeping these common pitfalls in mind, you’ll significantly reduce the chances of making errors in your Lewis structure drawings.
Advanced Tips for Mastering Lewis Structures
Once you’re comfortable with the basic structure of the sulfate ion, here are some advanced tips to refine your skills:
- Practice with Isoelectronic Species: Understanding structures like SO₃²⁻ and SO₃ can help deepen your comprehension of sulfur bonding. They follow similar patterns but with fewer oxygen atoms.
- Use Resonance Structures: Learn to apply resonance to complex ions. While sulfate doesn’t have resonance in the classical sense, understanding how to distribute formal charges correctly will enhance your ability to tackle more complex molecules.
- Visualize 3D Structures: Finally, try to visualize the 3D structure of sulfate using the Lewis structure. This will help reinforce your understanding of molecular geometry and how Lewis structures predict molecular shape.
With this comprehensive guide, you should now have a solid foundation for drawing the sulfate ion’s Lewis structure and understand how to avoid common mistakes. Practice these steps and tips, and soon you’ll master not only the sulfate ion but also other complex molecular structures.