Are you finding it tough to grasp the chemistry behind the reaction of strong acids and strong bases? Worry not, as we’re diving headfirst into this topic to unravel the complexities with clear, actionable advice and real-world examples. This guide aims to demystify the process, offering you tips, best practices, and problem-solving strategies to conquer your chemistry fears and understand these reactions comprehensively.
Understanding the Basics: Strong Acid and Strong Base Reactions
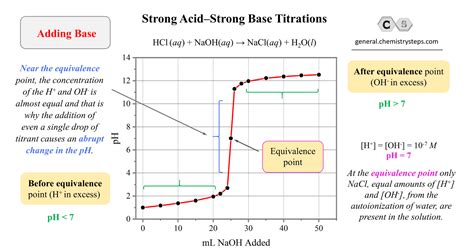
The reaction between a strong acid and a strong base is a classic example of an acid-base neutralization reaction. This reaction occurs when an acidic solution, characterized by a high concentration of hydrogen ions (H+), combines with a basic solution, characterized by a high concentration of hydroxide ions (OH-). The most notable outcome of this reaction is the formation of water and a salt.
Why It Matters
This type of reaction is fundamental in both chemistry and everyday life. Understanding it helps in fields like environmental science, medicine, and even cooking, where pH balance is crucial.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: To achieve a neutral pH, ensure equal molar concentrations of acid and base are mixed.
- Essential tip with step-by-step guidance: Add base to acid slowly, stirring continuously to observe neutralization points.
- Common mistake to avoid with solution: Avoid adding acid to base in high concentrations; it can cause a violent reaction.
Detailed How-To Section: Preparing for the Reaction
To delve deeper into this fascinating reaction, let’s walk through the process of preparing and observing a strong acid-strong base reaction. Follow these steps for a safe and educational experience.
Step 1: Choosing the Acid and Base
Select a strong acid like hydrochloric acid (HCl) or sulfuric acid (H2SO4) and a strong base like sodium hydroxide (NaOH) or potassium hydroxide (KOH). These are easily available and used in various laboratory settings.
Example: In this guide, we’ll use HCl (a strong acid) and NaOH (a strong base) for our reactions.
Step 2: Safety Precautions
Safety is paramount. Always wear protective gear, including gloves, goggles, and lab coats. Work in a well-ventilated area, preferably under a fume hood.
Key Tip: Have a spill kit and eye wash station nearby.
Step 3: Measure the Solutions
Accurate measurement is crucial for successful neutralization. Use a pipette for precise volume measurements or a burette for titration.
Best Practice: Use a pipette to measure 50 mL of HCl and 50 mL of NaOH in separate graduated cylinders.
Step 4: Mixing the Solutions
Slowly add the base to the acid while stirring continuously. The rate of addition can impact the reaction’s observation.
Example: Slowly pour the NaOH solution into the HCl solution while stirring. Watch for the pH indicator to turn neutral (usually around pH 7).
Step 5: Observing the Reaction
Pay attention to the heat produced during the reaction, which indicates the exothermic nature of neutralization. The heat indicates the reaction’s progress and completeness.
Tip: Use a thermometer to measure the temperature rise during the reaction.
Detailed How-To Section: Conducting a Titration
Titration is an advanced technique that not only helps in understanding the reaction but also quantifies the concentration of the acid or base.
Step 1: Setup
Prepare a burette by rinsing it with a small amount of the base solution you’ll be using. Fill it with NaOH and record the initial volume.
Best Practice: Ensure the burette is properly rinsed to avoid contamination.
Step 2: Add the Acid
Place a known volume of HCl (for example, 50 mL) in an Erlenmeyer flask. Add a few drops of a pH indicator, such as phenolphthalein, which turns pink in basic solutions.
Note: For safety, do not exceed 100 mL in a single flask.
Step 3: Titrate
Begin slowly adding the NaOH solution from the burette to the HCl in the flask, swirling the flask continuously. Watch the color change closely.
Key Tip: The endpoint is reached when the solution changes color and remains so even after a few swirls.
Step 4: Record the Volume
Record the final volume of the NaOH solution in the burette when the endpoint is reached. Use this volume to calculate the exact amount of NaOH added.
Calculation Example: If you started with 50 mL of HCl and added 25 mL of NaOH to reach the endpoint, the volume of NaOH is 25 mL.
Step 5: Analyze the Results
Use the volume of NaOH and its concentration to determine the concentration of the HCl. This calculation involves a straightforward application of the formula:
Formula: M_1V_1 = M_2V_2
Where: - ( M_1 ) and ( V_1 ) are the molarity and volume of the acid (HCl) - ( M_2 ) and ( V_2 ) are the molarity and volume of the base (NaOH)
Example: If M_2 (NaOH) is 1 M and V_2 is 25 mL, then to find M_1 (HCl), rearrange the formula to M_1 = \frac{M_2V_2}{V_1}
Practical FAQ
What if I observe an exothermic reaction becoming too vigorous?
If the reaction becomes too vigorous, immediately reduce the addition rate of the base to the acid to control the heat release. Always add the base slowly and cautiously, especially if working with larger volumes or higher concentrations. For a safe experience, you can also dilute the acid or base to lower their concentrations.
Can this reaction be reversed? How?
Neutralization reactions like this one are typically irreversible under normal conditions. However, in theory, if you could remove both the heat and the water formed, you might theoretically reverse the reaction by evaporating the water and applying high temperatures and pressures. But this isn’t practical in standard lab conditions.
Why does the pH indicator change color?
The pH indicator changes color because it is an acid-base indicator that reacts differently under acidic or basic conditions. When the base neutralizes the acid, the pH of the solution rises, shifting the color of the indicator as the solution transitions from acidic to neutral. For instance, phenolphthalein starts as colorless in acidic solutions and turns pink as the pH rises into the basic range.
Conclusion
Understanding the reaction between strong acids and strong bases not only deepens your grasp of fundamental chemistry but also equips you with practical knowledge applicable in many real-world scenarios. From environmental controls to medicinal formulations, mastering these reactions can significantly enhance your scientific proficiency. Always remember to approach these experiments with care and precision, using the detailed steps and tips provided to ensure a safe and educational experience.
By integrating these guidelines, tips, and examples into your study routine, you’re well on your way to mastering the nuances of acid-base reactions. Happy experimenting!