Strong Acid vs. Strong Base: Ultimate Chemistry Showdown
Welcome to the ultimate chemistry showdown where we dissect the intriguing battle between strong acids and strong bases. Understanding the fundamental differences, uses, and interactions between these chemical agents is crucial for both students and professionals in the field. In this guide, we will provide step-by-step guidance, practical examples, and expert tips to help you navigate the chemistry of acids and bases effectively.
One of the primary pain points for many users is figuring out how to differentiate between acids and bases, especially when it comes to handling them safely and understanding their interactions. This guide aims to demystify these processes and offer practical solutions to ensure you can confidently manage any acid-base situation.
Quick Reference
Quick Reference
- Immediate action item: Neutralize spills of strong acids or bases immediately using water to dilute and appropriate neutralizing agents to prevent harm.
- Essential tip: When diluting acids or bases, always add acid to water, never water to acid, to prevent exothermic reactions that could cause splashing.
- Common mistake to avoid: Failing to wear proper protective equipment like gloves and goggles when handling acids and bases can lead to serious injuries.
Understanding Strong Acids
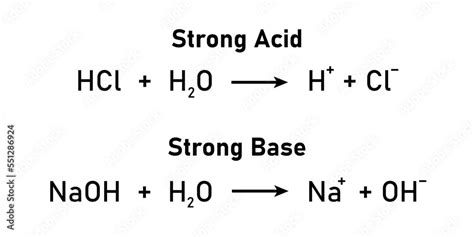
Strong acids completely dissociate in water, releasing a high concentration of hydrogen ions (H+). Examples include hydrochloric acid (HCl), sulfuric acid (H2SO4), and nitric acid (HNO3). These acids have significant uses in various industries, from manufacturing to laboratory experiments.
To effectively manage and utilize strong acids, follow these guidelines:
Identifying Strong Acids
To identify strong acids, look for the following characteristics:
- Complete dissociation in water
- High concentration of hydrogen ions
- Presence of hydrogen in the molecular formula
Always remember that strong acids are corrosive and can cause severe damage if not handled with care.
Handling Strong Acids Safely
Follow these steps to handle strong acids safely:
- Use personal protective equipment (PPE): Always wear gloves, goggles, and lab coats when handling strong acids.
- Avoid contact with skin and eyes: Strong acids can cause burns. If contact occurs, immediately rinse with plenty of water and seek medical attention.
- Proper storage: Store strong acids in tightly sealed containers away from incompatible substances (e.g., bases).
- Neutralize spills: If a strong acid spills, immediately dilute it with water and use appropriate neutralizing agents to prevent harm.
Neutralizing Strong Acids
When dealing with strong acids, it’s often necessary to neutralize them. Follow these steps:
- Dilute the acid: Gradually add acid to water to prevent exothermic reactions that could cause splashing.
- Neutralize: Use a strong base (like sodium hydroxide) to neutralize the acid. Always add the base to the acid slowly and with constant stirring.
- Monitor pH: Continuously monitor the pH of the solution to ensure the neutralization process is complete.
Understanding Strong Bases
Strong bases completely dissociate in water, releasing a high concentration of hydroxide ions (OH-). Examples include sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca(OH)2). Strong bases are used in a variety of applications, including cleaning, manufacturing, and laboratory settings.
To effectively manage and utilize strong bases, follow these guidelines:
Identifying Strong Bases
To identify strong bases, look for the following characteristics:
- Complete dissociation in water
- High concentration of hydroxide ions
- Presence of alkali metals or other strong base elements
Always remember that strong bases are highly corrosive and can cause severe damage if not handled with care.
Handling Strong Bases Safely
Follow these steps to handle strong bases safely:
- Use personal protective equipment (PPE): Always wear gloves, goggles, and lab coats when handling strong bases.
- Avoid contact with skin and eyes: Strong bases can cause burns. If contact occurs, immediately rinse with plenty of water and seek medical attention.
- Proper storage: Store strong bases in tightly sealed containers away from incompatible substances (e.g., acids).
- Neutralize spills: If a strong base spills, immediately dilute it with water and use appropriate neutralizing agents to prevent harm.
Neutralizing Strong Bases
When dealing with strong bases, it’s often necessary to neutralize them. Follow these steps:
- Dilute the base: Gradually add base to water to prevent exothermic reactions that could cause splashing.
- Neutralize: Use a strong acid (like hydrochloric acid) to neutralize the base. Always add the acid to the base slowly and with constant stirring.
- Monitor pH: Continuously monitor the pH of the solution to ensure the neutralization process is complete.
Practical FAQ
How do I determine if an acid is strong or weak?
To determine if an acid is strong or weak, consider the following:
- Strong acids completely dissociate in water, releasing a high concentration of hydrogen ions (H+). Examples include HCl, H2SO4, and HNO3.
- Weak acids partially dissociate in water, releasing a lower concentration of hydrogen ions. Examples include acetic acid (CH3COOH) and hydrogen fluoride (HF).
- Another way to identify strong acids is by their high Ka (acid dissociation constant) values. Strong acids typically have Ka values greater than 1.
Remember, strong acids are more corrosive and should be handled with extra care.
What happens when you mix a strong acid and a strong base?
When you mix a strong acid and a strong base, a neutralization reaction occurs, resulting in the formation of water and a salt. For example, mixing hydrochloric acid (HCl) with sodium hydroxide (NaOH) results in the formation of water (H2O) and sodium chloride (NaCl).
The general reaction can be written as:
HCl (aq) + NaOH (aq) → H2O (l) + NaCl (aq)
This reaction releases heat, so it’s crucial to add the acid to water slowly to prevent exothermic reactions that could cause splashing.
How can I safely dispose of leftover strong acids or bases?
To safely dispose of leftover strong acids or bases, follow these steps:
- Neutralize the chemical: If possible, neutralize the acid or base with an appropriate agent (e.g., adding acid to water for bases, adding base to water for acids).
- Use appropriate containers: Transfer the neutralized solution to an appropriate, labeled, and tightly sealed container.
- Consult local regulations: Follow local regulations for hazardous waste disposal. Contact a hazardous waste disposal service if unsure.
- Avoid down the drain: Do not dispose of strong acids or bases down the drain as it can cause severe environmental damage.
Following these steps ensures that you handle chemical waste safely and responsibly.
Conclusion
Mastering the handling,