Understanding the silver atomic number unlocks numerous scientific and practical insights. With an atomic number of 47, silver stands out as a versatile and historically significant element. This article delves into silver’s atomic structure, explores its practical applications, and reveals its hidden secrets based on expert perspectives and evidence-based statements.
The Science Behind Silver’s Atomic Number
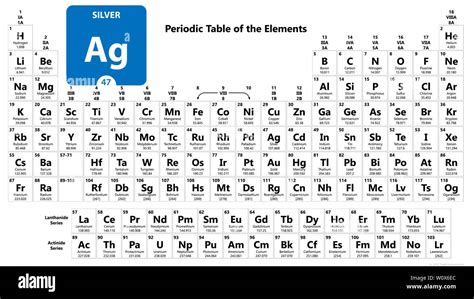
Silver’s atomic number 47 places it in Group 11 of the periodic table, making it a transition metal with a unique blend of properties. This atomic configuration dictates that silver has 47 protons in its nucleus and, in its neutral state, 47 electrons. These electrons are distributed in four main energy levels, which are pivotal in defining silver’s remarkable conductivity, malleability, and luster.
Silver’s electron configuration is [Kr] 4d10 5s1. The filled 4d subshell and the lone electron in the 5s orbital are key to its superior electrical and thermal conductivity, which are often used in electronics and medical devices. This unique atomic arrangement also results in silver’s characteristic reflective quality, making it indispensable in mirrors and mirrors-based technologies.
Practical Applications of Silver
The applications of silver extend across various industries, leveraging its atomic number properties for diverse practical uses. In the medical field, silver nanoparticles are widely used for their antimicrobial properties. The antimicrobial effect is due to silver’s ability to disrupt the cellular processes of bacteria, making it an effective agent in wound dressings and medical implants.
In the tech industry, silver’s conductivity is unmatched. It is a preferred material for high-performance electrical contacts, conductors, and photovoltaic cells. Due to its superior conductive properties, silver is also extensively used in the production of silver-plated wire and cables, ensuring efficient electrical transmission and minimizing resistance.
Beyond its industrial applications, silver plays a crucial role in everyday life. From jewelry to coins, silver’s aesthetic appeal and durability make it a favorite choice. Its unique luster and resistance to tarnishing mean it can maintain its beauty and value over time, making it a preferred material for high-quality silverware and decorative items.
Key Insights
- Primary insight with practical relevance: Silver’s atomic number 47 provides a framework for understanding its unparalleled conductivity and antimicrobial properties.
- Technical consideration with clear application: The electron configuration of silver enables its use in cutting-edge medical and electronic technologies.
- Actionable recommendation: Researchers and industry professionals should explore the use of silver nanoparticles in new product development for enhanced performance and functionality.
Is silver’s conductivity better than copper?
Yes, silver has a higher electrical conductivity compared to copper, making it preferred in applications requiring maximum efficiency and minimal resistance.
Can silver’s antimicrobial properties be harnessed for new medical applications?
Absolutely. Silver’s strong antimicrobial properties can be utilized in creating advanced wound care products, coatings for medical devices, and other innovative healthcare solutions.
Understanding silver’s atomic number 47 not only highlights its intrinsic properties but also its wide-ranging applications that drive technological and medical advancements. Whether in the realm of electronics, healthcare, or luxury goods, silver’s unique attributes continue to unveil hidden secrets and unlock new possibilities.