If you're navigating the intricate world of Regents Chemistry, you’re likely seeking a path that leads to your success on the exam. This guide is tailored to address the unique challenges you face while providing step-by-step guidance, actionable advice, and practical solutions that will empower you to master the course material.
From understanding complex chemical reactions to interpreting laboratory data, the road to mastering Regents Chemistry can seem daunting. However, armed with the right strategies and persistent effort, you can transform your approach and achieve top performance. This guide will walk you through essential concepts, offer real-world examples, and highlight common pitfalls to help you avoid them.
Understanding Your Study Philosophy
To excel in Regents Chemistry, it’s crucial to develop a clear and effective study philosophy. Start by breaking down your learning objectives into manageable parts, focusing on understanding rather than rote memorization. Prioritize topics that are more challenging, and allocate time for practice problems and lab simulations to reinforce your knowledge.
Incorporate active learning techniques, such as teaching the material to a peer or discussing concepts in study groups. This not only enhances your comprehension but also prepares you for the exam’s diverse question formats.
Quick Reference
Quick Reference
- Immediate action item: Begin your study sessions by reviewing the core concepts you’ve previously learned. This foundational knowledge will make new material more approachable.
- Essential tip: Create a schedule that balances theory and practical applications, including regular review sessions and problem-solving practice.
- Common mistake to avoid: Neglecting lab work practice. Labs are a significant part of the exam and understanding them is vital for success.
Mastering Chemical Reactions
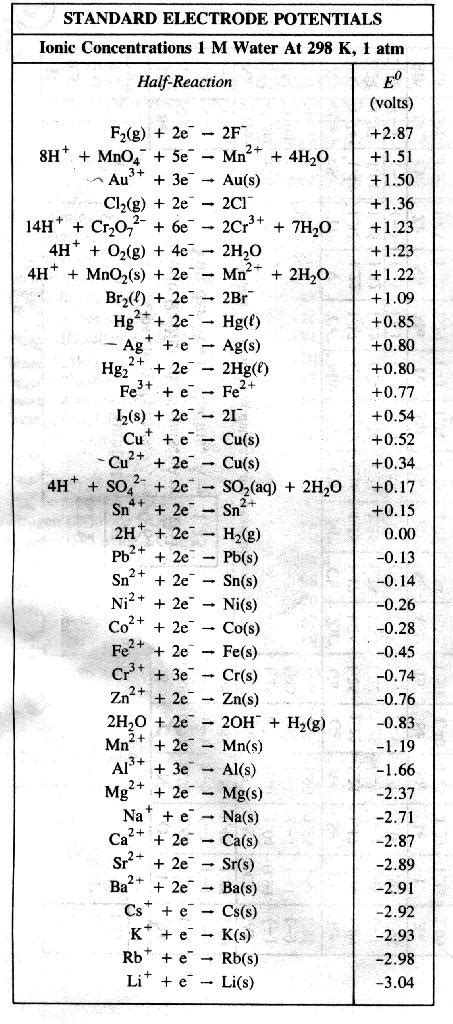
Understanding chemical reactions is a cornerstone of Regents Chemistry. Let’s dive into the fundamental types of chemical reactions, with a focus on how to recognize and apply these concepts effectively.
Types of Chemical Reactions
Chemical reactions can generally be categorized into several types:
- Synthesis: Two or more reactants combine to form a single product (e.g., A + B → AB).
- Decomposition: A single compound breaks down into two or more simpler substances (e.g., AB → A + B).
- Single Displacement: An element reacts with a compound, and an element from the compound is displaced (e.g., A + BC → AC + B).
- Double Displacement: Two compounds exchange parts, forming two new compounds (e.g., AB + CD → AD + CB).
- Combustion: A substance reacts with oxygen, releasing energy in the form of heat and light (e.g., C + O2 → CO2).
Step-by-Step Guide to Balancing Chemical Equations
Balancing chemical equations is a fundamental skill that requires practice to master. Follow these steps:
- Write the unbalanced equation: Start by writing the chemical equation with the reactants and products listed.
- Identify the number of atoms: Count the number of each type of atom on both sides of the equation.
- Balance one type of atom at a time: Start with the most complex molecule and balance it first, followed by simpler molecules.
- Use coefficients: Place whole number coefficients in front of the chemical formulas to balance the atoms.
- Check your work: Verify that the number of each type of atom is the same on both sides of the equation.
For example, to balance the combustion of propane (C3H8):
| Unbalanced equation: | C3H8 + O2 → CO2 + H2O |
| Balance carbon atoms: | C3H8 + O2 → 3CO2 + H2O |
| Balance hydrogen atoms: | C3H8 + O2 → 3CO2 + 4H2O |
| Balance oxygen atoms: | C3H8 + 5O2 → 3CO2 + 4H2O |
By following these steps, you can systematically balance complex chemical equations.
Practical Application: Laboratory Experiment
To solidify your understanding, let’s consider a practical lab experiment where you observe a synthesis reaction:
Suppose you are tasked with synthesizing a simple salt by reacting sodium chloride (NaCl) with sulfuric acid (H2SO4).
Here’s how you proceed:
- Setup: Place an appropriate amount of sodium (Na) in a crucible and heat it using a Bunsen burner until it forms sodium chloride (NaCl).
- Combine: Combine NaCl with an appropriate amount of sulfuric acid (H2SO4) in a beaker.
- Observe: Watch as the reaction takes place, noting any changes in temperature, color, or gas production.
- Record: Document your observations and the balanced chemical equation:
| Chemical equation: | Na + H2SO4 → NaCl + H2O + SO2 |
FAQ Section
What should I do if I struggle with understanding stoichiometry?
Struggling with stoichiometry is common, but it’s definitely conquerable with practice. Here’s what you can do:
- Review the basics: Make sure you understand the fundamental concepts of moles, molar mass, and the mole ratio.
- Practice problems: Work through a variety of stoichiometry problems, starting with simple ones and gradually moving to more complex examples.
- Use visual aids: Drawing diagrams or using models can help visualize the relationships between reactants and products.
- Seek help: If you’re still struggling, don’t hesitate to ask for help from your teacher, a tutor, or a study group.
Remember, stoichiometry requires both logical reasoning and a good understanding of basic chemistry principles, so the more you practice, the more intuitive it will become.
Advanced Techniques in Chemical Calculations
As you become more proficient, delve into advanced techniques in chemical calculations. Here, you will explore methods for solving more intricate stoichiometric problems and handling advanced lab scenarios.
Using Limiting Reactants in Stoichiometry
Identifying and calculating the limiting reactant is essential in stoichiometry. This is the reactant that will be entirely consumed first in a reaction, thereby limiting the amount of product formed.
To determine the limiting reactant, follow these steps:
- Write the balanced chemical equation: Start with the balanced equation for the reaction.
- Convert reactants to moles: Use the molar mass to convert the given amounts of reactants to moles.
- Calculate moles of product