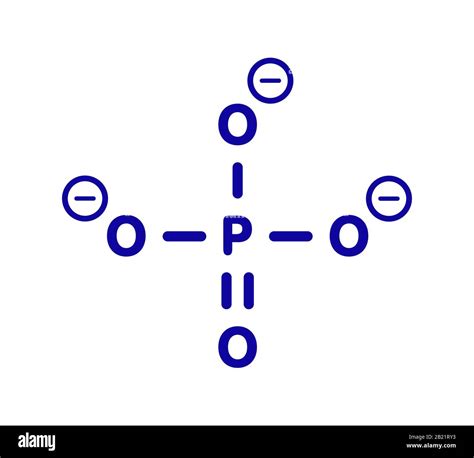
Mastering the Phosphate Lewis Structure: Key Insights Unveiled
Understanding the phosphate ion (PO₄³⁻) is crucial for anyone delving into advanced chemistry. The Lewis structure of phosphate, a central aspect of its bonding and properties, can initially appear complex but becomes much clearer when broken down step-by-step. This guide provides actionable advice, real-world examples, and practical solutions to help you fully grasp this fundamental concept.
Problem-Solution Opening Addressing User Needs
Many students find the phosphate Lewis structure a daunting task, often because they lack a clear, step-by-step approach or struggle to visualize electron distributions. This guide is designed to demystify the process and arm you with the tools to master this concept confidently. Our goal is to address your specific pain points, ensuring that you not only understand the structure but can apply this knowledge to other complex molecular geometries. Whether you're prepping for an exam or exploring advanced chemistry topics, this guide will equip you with the insights needed to succeed.
Quick Reference
- Immediate action item with clear benefit: Start by counting the total valence electrons needed for the PO₄³⁻ ion.
- Essential tip with step-by-step guidance: Place phosphorus in the center and surround it with four oxygen atoms, connecting them with single bonds. Distribute the remaining electrons to satisfy the octet rule for each atom.
- Common mistake to avoid with solution: Do not forget to account for the -3 charge on the phosphate ion; it will affect how you distribute your electrons and the overall stability of the structure.
Step-by-Step Guide to Drawing the Phosphate Lewis Structure
Drawing a Lewis structure involves several steps, each with its own set of rules and guidelines. Here's a detailed breakdown for the phosphate ion, ensuring you understand each part thoroughly.
Step 1: Count Valence Electrons
The first step is to determine the total number of valence electrons you need. Remember that phosphorus (P) is in group 15 and has 5 valence electrons, while each oxygen (O) atom, in group 16, has 6 valence electrons. Additionally, the phosphate ion carries a -3 charge, meaning we must account for 3 extra electrons.
- Phosphorus: 5 valence electrons
- Each oxygen: 6 valence electrons × 4 oxygens = 24 valence electrons
- Extra electrons for the -3 charge: 3 electrons
- Total: 5 + 24 + 3 = 32 valence electrons
Step 2: Structure Layout
The next step is to place phosphorus at the center, surrounded by four oxygen atoms. Connect each oxygen atom to phosphorus with a single bond. Each bond represents two electrons.
- Single bond between phosphorus and each oxygen: 4 bonds × 2 electrons = 8 electrons
- After bonding, we have: 32 - 8 = 24 electrons left
Step 3: Complete Octets
Now, distribute the remaining electrons to satisfy the octet rule for each atom. Each oxygen atom already has a complete octet with the initial single bonds, so we'll focus on phosphorus.
- Phosphorus needs 8 electrons to complete its octet.
- Each oxygen atom will share any remaining electrons to achieve a full octet if possible.
Since there are 24 electrons left after the initial bond formation, we distribute them to phosphorus first, then to oxygens as necessary:
- Add 8 electrons to phosphorus to complete its octet.
- This uses up 8 electrons, leaving us with 16.
- Distribute the remaining 16 electrons in pairs among the four oxygen atoms, giving each oxygen an additional lone pair and thus completing their octets.
Step 4: Check the Structure
Finally, check to ensure the structure is stable. Verify that all atoms fulfill the octet rule, and ensure the overall charge is -3. Any leftover electrons will be placed on the phosphorus atom if necessary to achieve the -3 charge on the ion.
Tips, Best Practices, and How-to Information
Here are some additional tips and best practices to keep in mind while drawing Lewis structures for other ions and molecules:
- Prioritize Central Atom Placement: Choose the least electronegative atom as the central atom, usually, this will be phosphorus in phosphate.
- Always Count Electrons: Double-check the total number of valence electrons for accuracy. Mistakes in counting can lead to structural errors.
- Manage Charges Carefully: Always account for the overall charge of the ion when distributing electrons.
- Distribute Electrons Wisely: Start by giving lone pairs to the most electronegative atoms (oxygens in this case) and work outward to phosphorus.
- Check for Resonance: If possible, consider resonance structures to achieve a more stable Lewis configuration.
Practical FAQ
Why does the phosphate ion have a -3 charge?
The phosphate ion (PO₄³⁻) has a -3 charge because it has gained three extra electrons beyond its neutral atomic form. Phosphorus naturally has 5 valence electrons, and when bonded to four oxygen atoms, each oxygen needs two electrons to complete its octet. However, since oxygen typically has 6 valence electrons and only needs 2 to bond with phosphorus, the extra electrons come from outside the molecule. These three extra electrons contribute to the -3 charge on the phosphate ion.
What happens if I don’t account for the phosphate ion’s charge?
If you don’t account for the -3 charge of the phosphate ion when drawing the Lewis structure, your final structure will be incorrect. For instance, you might complete the octets for phosphorus and oxygen atoms without considering the extra electrons, leading to an incorrect total number of electrons and a charge that doesn’t match the -3 charge. This oversight can significantly alter the stability and properties of the molecule as predicted.
Can the phosphate ion form resonance structures?
Yes, the phosphate ion can form resonance structures. In a resonance structure, the electrons in the bonds between phosphorus and oxygen atoms are shared, allowing the bonding electrons to move to different oxygen atoms. This delocalization of electrons helps distribute the formal charge more evenly and stabilizes the overall structure. Each resonance structure will have single bonds between phosphorus and three of the oxygen atoms and a double bond with the fourth oxygen, maintaining a total of 8 electrons around phosphorus and a -3 charge overall.
Conclusion
Mastering the phosphate Lewis structure involves understanding the principles of valence electrons, central atom placement, and charge accounting. By following the step-by-step guide and utilizing the best practices shared in this guide, you will be well-equipped to tackle not just the phosphate ion, but other complex molecules as well. Remember, practice is key—the more you apply these concepts, the more intuitive they will become. Happy learning!