Understanding the PCl3 Lewis structure is essential for grasping fundamental concepts in molecular chemistry. Phosphorous trichloride (PCl3) is a crucial compound in organic synthesis, known for its role as a chlorinating agent. This article delves into the nuances of its Lewis structure, providing an insightful and evidence-based exploration.
Key Insights
- PCl3 has a trigonal pyramidal molecular geometry.
- It features phosphorus with a lone pair, impacting its polarity.
- Proper drawing of the Lewis structure is vital for understanding its chemical behavior.
Understanding the Basics of PCl3
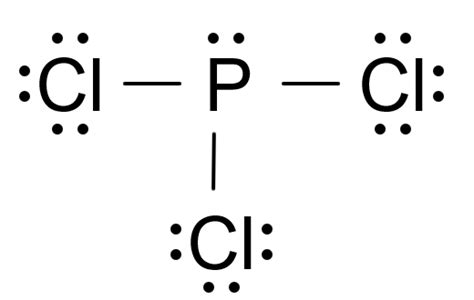
To grasp the PCl3 Lewis structure, we must first understand its atomic composition. The molecule consists of one phosphorus atom bonded to three chlorine atoms. Phosphorus, a member of the nitrogen group, has five valence electrons, while each chlorine atom has seven. Understanding the electron configuration and bonding within PCl3 is fundamental to forming an accurate Lewis structure.
A Lewis structure represents how atoms in a molecule are bonded together, including lone pairs of electrons. In PCl3, phosphorus forms three single bonds with three chlorine atoms, utilizing three of its valence electrons. The remaining two valence electrons on phosphorus form a lone pair, leading to a trigonal pyramidal molecular geometry. This arrangement ensures that the lone pair remains as far as possible from the bonding electrons, minimizing electron-electron repulsion.
Drawing the PCl3 Lewis Structure
The creation of the PCl3 Lewis structure involves several deliberate steps to ensure accuracy and completeness. To draw it, start by placing phosphorus in the center, surrounded by three chlorine atoms.
- Start by counting the total number of valence electrons: 5 from phosphorus plus 3*7 from chlorine atoms, totaling 26 electrons.
- Distribute these electrons in pairs to form the phosphorus-chlorine bonds, using 6 electrons (3 pairs).
- Place the remaining electrons around the central atom. After bonding, phosphorus uses 2 of its valence electrons to form a lone pair, using up 2 more electrons. The remaining 18 electrons (9 pairs) are distributed to chlorine atoms, each getting 3 pairs.
- Reassess the structure. Verify that octets are satisfied for chlorine atoms, and phosphorus has an expanded octet due to its ability to accommodate more than eight electrons.
The final structure shows phosphorus with a lone pair and three single bonds to chlorine atoms, leading to a trigonal pyramidal shape. This Lewis structure allows chemists to predict chemical reactivity and molecular properties accurately.
Why is the PCl3 molecule polar?
The lone pair on the phosphorus atom creates an uneven charge distribution, making the molecule polar. This polarity is essential in explaining its solubility in organic solvents and its reactivity.
What are the implications of PCl3's geometry?
The trigonal pyramidal geometry of PCl3 affects its reactivity and intermolecular forces. It enables the molecule to interact more effectively with other molecules, contributing to its use in synthetic chemistry.
Mastering the PCl3 Lewis structure deepens our understanding of its chemical properties and applications. With a clear structure, we can accurately predict its behavior in various chemical environments. Such detailed knowledge is invaluable for anyone engaged in chemical research and industrial applications.