Are you finding it challenging to understand the intricate details of NF3 molecular geometry and its implications on bonding? If so, you’re not alone. Many students and even professionals encounter difficulties grasping the specific configurations and bonding in molecules like NF3. This guide will provide a comprehensive, step-by-step breakdown of NF3 molecular geometry, with actionable advice, real-world examples, and a conversational expert tone to ensure clarity and accessibility. Let’s dive in and demystify the shape and bonding of NF3.
Understanding NF3 Molecular Geometry: Step-by-Step Guidance
NF3, or nitrogen trifluoride, is a fascinating molecule often encountered in advanced chemistry courses. Its molecular geometry plays a critical role in its chemical behavior, reactivity, and even physical properties. To get a grasp on NF3 molecular geometry, we need to consider several aspects: the arrangement of atoms around the central nitrogen atom, the bond angles, and the type of bonding present.
Quick Reference Guide to NF3
Quick Reference
- Immediate action item with clear benefit: Always start with Lewis structure to determine the shape of the molecule.
- Essential tip with step-by-step guidance: Count valence electrons and distribute them appropriately to satisfy the octet rule.
- Common mistake to avoid with solution: Misinterpreting bond angles due to lack of understanding of VSEPR theory.
This quick reference will help you keep the essential points in mind while studying NF3 molecular geometry.
Breaking Down the Lewis Structure of NF3
The first step in understanding NF3 molecular geometry is drawing its Lewis structure. This visual representation is crucial for identifying the basic shape and understanding the distribution of electrons in the molecule.
Here’s how to do it:
1. Start by identifying the total number of valence electrons. Nitrogen has 5, and each fluorine has 7. For three fluorines, this sums up to:
5 (from nitrogen) + 3 × 7 (from fluorines) = 26 valence electrons
2. Place the least electronegative atom at the center. Here, nitrogen will be the central atom. Then, arrange the three fluorines around it.
3. Connect the central atom to the surrounding atoms with single bonds. Each bond uses up 2 electrons, so for three bonds, 6 electrons are used.
4. Distribute the remaining electrons to satisfy the octet rule for all atoms. Remember, fluorines are happy with 8 electrons, but nitrogen may need an exception:
Remaining electrons = 26 - 6 = 20 electrons.
5. To satisfy the octet rule on nitrogen, place three lone pairs on it, using up 6 electrons:
Remaining electrons = 20 - 6 = 14 electrons.
6. Distribute the remaining 14 electrons in pairs to the three fluorines, ensuring each gets 3 lone pairs (6 electrons). Now, nitrogen has one lone pair and three single bonds with fluorine atoms.
By following these steps, you can derive the Lewis structure for NF3.
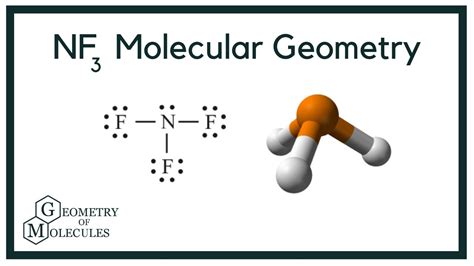
The resulting structure shows nitrogen at the center with three single bonds to fluorines and one lone pair, giving it a trigonal pyramidal shape.
Deciphering NF3 Molecular Shape
Understanding the molecular shape is crucial for predicting the molecule’s behavior. For NF3:
1. Utilize the VSEPR (Valence Shell Electron Pair Repulsion) theory. This theory states that electron pairs around a central atom repel each other and arrange themselves to be as far apart as possible.
2. Count both bonding pairs and lone pairs around the central atom:
Nitrogen has 3 bonding pairs with fluorine and 1 lone pair.
3. Predict the shape based on the total electron pairs:
The presence of 4 electron pairs (3 bonding + 1 lone) leads to a tetrahedral electron-pair geometry.
However, the molecular shape is dictated by the positions of the atoms only, so NF3 adopts a trigonal pyramidal shape because the lone pair takes up more space and alters the bond angles slightly.
Detailed How-to: Analyzing Bond Angles in NF3
Bond angles are vital for understanding the molecular geometry and predicting chemical interactions. In NF3, the bond angle between the fluorine atoms is slightly less than 109.5 degrees due to the repulsion from the lone pair on nitrogen.
Here’s a detailed process to understand bond angles:
1. Recall that in a perfect tetrahedral arrangement, the bond angle would be 109.5 degrees. This is due to the spatial arrangement that minimizes repulsion between all electron pairs.
2. Consider the impact of lone pairs. In NF3, the lone pair on nitrogen occupies more space than bonding pairs because it is closer to the nitrogen atom. This results in increased repulsion with the bonding pairs, leading to slightly reduced bond angles.
3. Measure or estimate the bond angles using molecular modeling software or reference tables. In NF3, the F-N-F bond angle is around 102 degrees.
By understanding how lone pairs affect the angles, you can predict and explain the molecule’s three-dimensional shape.
Practical FAQ on NF3 Molecular Geometry
Why does NF3 have a trigonal pyramidal shape instead of tetrahedral?
In a perfect tetrahedral arrangement, all four electron pairs would be equally spaced, leading to bond angles of 109.5 degrees. However, NF3 has one lone pair and three bonding pairs. The lone pair repels the bonding pairs more strongly due to its closer proximity to the central nitrogen atom, causing the bond angles to decrease slightly. This repulsion makes the molecular shape trigonal pyramidal rather than tetrahedral.
Understanding why NF3 adopts a trigonal pyramidal shape is crucial for grasping its molecular geometry fully.
Best Practices for Learning NF3 Geometry
To master the geometry of NF3 and similar molecules, consider these best practices:
- Start with the Lewis structure to understand electron distribution.
- Use VSEPR theory to predict the shape and bond angles.
- Visualize the molecule using molecular models or software to grasp its three-dimensional nature.
- Practice drawing different molecules to build proficiency in identifying shapes and bond angles.
- Refer to reliable sources and consult additional materials if stuck.
By incorporating these practices, you will reinforce your understanding and gain confidence in tackling NF3 and other complex molecules.
Remember, the key to mastering molecular geometry lies in practice and a solid grasp of foundational concepts like the Lewis structure and VSEPR theory. This guide should serve as a solid foundation to build on your knowledge, enabling you to explore the fascinating world of molecular geometry confidently.