Are you a budding chemist or a curious student looking to understand the basics of chemical compounds? You’ve landed in the right place. This guide will help you uncover the secrets of the NF3 compound name, breaking down its components and exploring its characteristics in a way that’s easy to grasp and useful to apply. We’ll dive into the practical aspects, address common questions, and offer tips to avoid common pitfalls. Let’s embark on this chemistry journey together!
Understanding NF3: A Problem-Solution Approach
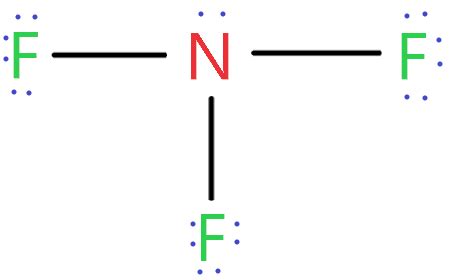
The NF3 compound name can initially seem complex, but fear not! By breaking it down into understandable parts, we can demystify this chemical term. NF3 refers to an inorganic compound composed of nitrogen (N) and fluorine (F). The “NF3” represents three fluorine atoms bonded to one nitrogen atom. Understanding this compound is crucial for grasping the basics of molecular chemistry, which will have applications in various scientific fields.
The challenge lies in not just identifying the components but also understanding the structure, reactivity, and uses of this compound. This guide will help you navigate these complexities, providing actionable advice, real-world examples, and problem-solving strategies to overcome any confusion you might face.
Quick Reference Guide
Quick Reference
- Immediate action item: Identify the elements in NF3. Nitrogen (N) is bonded to three fluorine (F) atoms.
- Essential tip: To understand its molecular geometry, use the VSEPR theory, which predicts a trigonal pyramidal shape due to the three fluorine atoms and one lone pair on the nitrogen atom.
- Common mistake to avoid: Confusing NF3 with other similar nitrogen and fluorine compounds like NFx. Ensure you’re counting the fluorine atoms correctly.
Deep Dive into NF3’s Structure and Bonding
Let’s begin with the fundamental building blocks of NF3. Nitrogen and fluorine have different electronegativities, creating a polar bond when they combine. Nitrogen, with its position in Group 15, can accommodate three electrons from three fluorine atoms, each forming a single bond.
Here’s a step-by-step breakdown of its structure:
- Identify the central atom: In NF3, nitrogen (N) is the central atom due to its lower electronegativity compared to fluorine.
- Count valence electrons: Nitrogen has 5 valence electrons, and each fluorine has 7. With three fluorine atoms, you get a total of 26 valence electrons.
- Distribute electrons: Place the valence electrons around the central atom, starting with nitrogen. Pair each fluorine atom with nitrogen, forming three single bonds, using up 6 electrons (3 pairs).
- Check remaining electrons: With 20 electrons left (26 total - 6 used), place three around each fluorine, using up 6 electrons, leaving 4 electrons. Place these 4 electrons on nitrogen to complete its octet, forming a lone pair.
- Molecular geometry: Using the VSEPR theory, the nitrogen atom surrounded by three bonding pairs and one lone pair adopts a trigonal pyramidal shape.
Understanding this structure helps predict its reactivity and physical properties. For example, its polarity due to the electronegativity difference between nitrogen and fluorine makes it a good candidate for various chemical reactions.
Practical Applications of NF3
NF3 has several practical applications across various industries:
1. Rocket Propellant: Due to its high oxidation potential, NF3 is used as an oxidizing agent in rocket propellants.
2. Semiconductor Industry: It acts as a fluorinating agent in the fabrication of semiconductor devices.
3. Chemical Synthesis: NF3 is used to introduce fluorine into organic compounds, which is essential for pharmaceuticals and agrochemicals.
Tips for Handling NF3
Given its reactivity and potential hazards, proper handling is crucial:
- Storage: Store NF3 in a cool, well-ventilated area away from strong bases, reducing the risk of unwanted reactions.
- Safety Gear: Always wear appropriate personal protective equipment (PPE) such as gloves, goggles, and a lab coat.
- Disposal: Dispose of NF3 waste according to local regulations to avoid environmental contamination.
FAQs on NF3 Usage and Handling
What is the primary use of NF3 in industry?
NF3 is primarily used in the aerospace industry as an oxidizing agent in rocket propellants due to its high oxidation potential. Additionally, it plays a crucial role in the semiconductor industry as a fluorinating agent for manufacturing semiconductor devices.
How should I store NF3 to ensure its stability?
NF3 should be stored in a cool, well-ventilated area. Keep the storage container tightly closed and away from sources of heat and open flames. It should also be kept away from incompatible materials such as strong bases, which can trigger unwanted reactions.
What precautions should I take when handling NF3?
When handling NF3, always prioritize safety. Wear appropriate personal protective equipment (PPE), including gloves, goggles, and a lab coat. Work in a well-ventilated area or under a fume hood to avoid inhalation of fumes. Follow all manufacturer-provided guidelines and local safety regulations.
Understanding NF3 is pivotal for both academic and industrial applications. By following the detailed guide, you’ll be equipped with the knowledge and practical steps needed to grasp this compound’s structure, reactivity, and uses. Always remember to approach its handling with the necessary safety precautions to mitigate any potential risks. Dive in and explore the fascinating world of chemistry!