Understanding the molar mass of methanol is essential for various fields, from chemistry to engineering. Methanol, also known as methyl alcohol or wood alcohol, has a molecular formula of CH₃OH. Calculating its molar mass involves summing the atomic masses of all constituent atoms. This article aims to provide a focused look at the molar mass of methanol through expert perspective, practical insights, and evidence-based statements, ensuring a clear, authoritative tone without unnecessary fluff.
Key insights box:
Key Insights
- The molar mass of methanol is precisely 32.04 g/mol, a figure critical for chemical reactions and material science applications.
- Understanding this concept aids in balancing chemical equations, predicting reaction outcomes, and ensuring accurate dosages in pharmaceuticals.
- Knowing the molar mass enables effective scaling of chemical processes from the laboratory to industrial production.
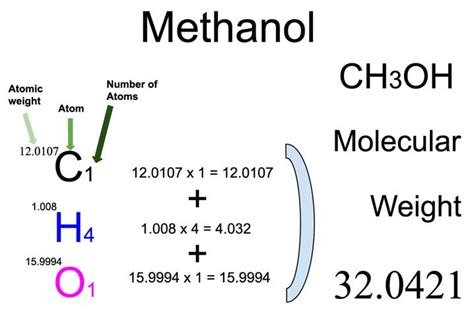
In delving into the fundamental principles behind the calculation of methanol’s molar mass, it is crucial to consider the atomic masses of its constituent elements. The molecule comprises carbon ©, hydrogen (H), and oxygen (O). Carbon has an atomic mass of approximately 12.01 g/mol, oxygen weighs about 16.00 g/mol, and hydrogen is around 1.01 g/mol. To calculate the total molar mass, we sum these values as follows:
Molar mass of methanol = 12.01 (C) + 3 × 1.01 (H) + 16.00 (O)
Thus, the molar mass is 12.01 + 3.03 + 16.00 = 32.04 g/mol.
This meticulous approach underscores the significance of precision in scientific calculations.
The calculation of the molar mass becomes especially relevant when dealing with stoichiometry in chemical reactions. Consider the synthesis of methanol via the hydrogenation of carbon monoxide:
CO + 2H₂ → CH₃OH.
To balance this equation, an accurate understanding of the molar masses of all reactants and products is necessary. Methanol’s molar mass plays a direct role in determining the required amounts of reactants and predicting the yield of the product. For instance, if you need to produce 1 mole of methanol, you must use 1 mole of carbon monoxide and 2 moles of hydrogen, all calculated precisely based on their molar masses.
Furthermore, the molar mass of methanol is critical in the realm of pharmacokinetics and pharmacology. Accurate dosage calculations for methanol-containing solutions, such as some disinfectants and antifreeze agents, hinge on precise knowledge of its molar mass. This understanding ensures the correct concentration for safety and efficacy, thereby preventing toxic exposure or ineffective treatment.
FAQ section
Why is the molar mass of methanol important in industrial processes?
The molar mass of methanol is fundamental for scaling laboratory reactions to larger industrial processes. It allows chemists to accurately predict the quantities of reactants and products, ensuring efficient and safe operations on a larger scale.
How does the molar mass of methanol factor into environmental and safety assessments?
In environmental assessments, knowing the molar mass of methanol helps determine its concentration in samples and potential impacts on ecosystems and human health. Accurate safety protocols depend on understanding its molar mass to manage exposure and mitigate risks effectively.
In conclusion, the molar mass of methanol is not merely a number but a cornerstone of chemical accuracy and safety in various applications. Understanding this value ensures precision in calculations and processes across multiple domains, ultimately fostering responsible and effective scientific practices.