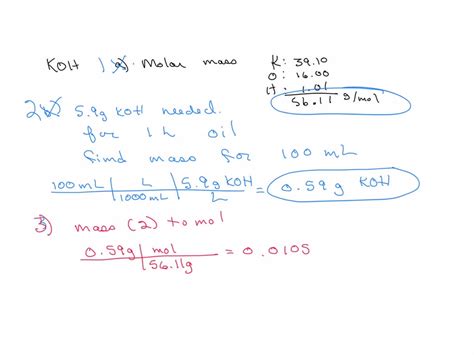Welcome to the comprehensive guide on understanding the molar mass of potassium hydroxide (KOH), a crucial concept in chemistry. The molar mass of KOH is not just a numeric value; it encapsulates a fundamental understanding of chemical composition, which is essential for anyone studying chemistry at any level. This guide is designed to help you grasp the essentials of molar mass with clear, actionable steps, practical examples, and problem-solving insights. Whether you are a student, a teacher, or simply a curious mind, this guide will serve as your go-to resource for mastering this aspect of chemistry.
Understanding the Problem: Why Molar Mass Matters
Calculating the molar mass of compounds is a foundational skill in chemistry that facilitates understanding of chemical reactions, stoichiometry, and the physical properties of substances. Many students struggle with the concept of molar mass because it often seems abstract and disconnected from real-world applications. Without a firm grasp of how to calculate molar mass, students may find it challenging to predict reaction outcomes and understand the relationships between different substances.
Our aim in this guide is to break down the concept of molar mass into manageable, understandable segments. We will provide step-by-step instructions, illustrate practical applications, and address common misconceptions to ensure you not only learn the theory but also see its relevance in everyday chemistry.
Quick Reference
Quick Reference
- Immediate action item: Calculate the molar mass of KOH by adding the atomic masses of potassium (K), oxygen (O), and hydrogen (H) on the periodic table.
- Essential tip: Use a reliable periodic table to ensure accuracy in atomic masses.
- Common mistake to avoid: Confusing molar mass with molecular mass; remember, molar mass is in grams per mole.
Detailed How-To Section: Calculating Molar Mass of KOH
Calculating the molar mass of KOH involves summing the atomic masses of its constituent elements. Let’s break this down step-by-step:
- Identify the elements: The formula KOH consists of potassium (K), oxygen (O), and hydrogen (H).
- Find atomic masses: Use a periodic table to find the atomic masses of each element:
Element Atomic Mass Potassium (K) 39.10 g/mol Oxygen (O) 16.00 g/mol Hydrogen (H) 1.01 g/mol - Sum the atomic masses: Add the atomic masses of K, O, and H together to get the molar mass of KOH.
Molar mass of KOH = 39.10 g/mol (K) + 16.00 g/mol (O) + 1.01 g/mol (H) = 56.11 g/mol
- Verify the result: Double-check your calculations to ensure there are no errors.
Understanding this calculation is crucial as it applies to various other chemical compounds and reactions. Mastery here will help you tackle more complex chemical problems.
Detailed How-To Section: Applying Molar Mass in Chemical Reactions
The molar mass of a compound like KOH is essential not just for individual calculations, but for understanding the proportions and relationships in chemical reactions. Let’s dive into how you can use this concept to balance chemical equations and determine reactant and product quantities.
Consider a scenario where you need to prepare a solution of KOH. Knowing the molar mass allows you to accurately measure the correct amount of KOH needed to achieve the desired concentration. Here’s how:
- Determine the desired concentration: Suppose you need a 1.0 M KOH solution. The molarity (M) is defined as the number of moles of solute per liter of solution.
Molar mass of KOH = 56.11 g/mol
Number of moles in 1.0 L of 1.0 M KOH solution = 1.0 mol/L * 1.0 L = 1.0 mol
- Calculate the mass needed: Use the molar mass to determine the mass of KOH required.
Mass of KOH = number of moles * molar mass
Mass of KOH = 1.0 mol * 56.11 g/mol = 56.11 g
- Prepare the solution: Accurately weigh out 56.11 g of KOH and dissolve it in water to make 1.0 L of solution.
This practical example illustrates the real-world application of molar mass calculations. From laboratory experiments to industrial processes, understanding molar mass is vital for precise and effective chemical work.
Practical FAQ: Common Queries About Molar Mass
What is the difference between molar mass and molecular mass?
Molar mass and molecular mass often get confused, but they refer to different concepts. Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol), and it applies to all substances, whether elements or compounds. Molecular mass, on the other hand, is the sum of the atomic masses of the atoms in a molecule and is typically expressed in atomic mass units (amu). For example, the molar mass of KOH is 56.11 g/mol, while its molecular mass is the same numerical value but considered in amu when calculating the mass of one molecule.
Why is it important to use accurate atomic masses?
Using accurate atomic masses is crucial for several reasons. First, it ensures the precision of your molar mass calculations, which in turn affects stoichiometric calculations in chemical reactions. Inaccurate atomic masses can lead to incorrect determinations of the amounts of reactants and products, potentially altering the outcomes of experiments and industrial processes. Moreover, precise calculations foster better understanding and application of chemical principles.
How can errors in molar mass calculations affect chemical reactions?
Errors in molar mass calculations can have significant repercussions in both laboratory and industrial settings. For instance, if you miscalculate the amount of KOH needed for a reaction based on an incorrect molar mass, the reaction may not proceed as expected. This can lead to incomplete reactions, waste of resources, or even safety hazards in some cases. Accurate molar mass calculations are fundamental to achieving desired reaction outcomes and maintaining consistency in chemical processes.
This guide has aimed to demystify the concept of molar mass through practical steps, real-world applications, and a focus on resolving common issues that arise. By following the instructions provided, you should feel more confident in calculating the molar mass of compounds like KOH and understanding its implications in chemical reactions.
Remember, chemistry is a detailed science that requires precision and a clear understanding of fundamental concepts. With this guide, you now have a solid foundation for exploring more complex topics and applying these principles to your studies or professional work. Happy calculating!