The molar mass of potassium chloride, or KCl, is a fundamental concept in chemistry that plays a significant role in understanding chemical reactions, stoichiometry, and the properties of compounds. This seemingly simple calculation has broad implications in various fields, from industrial manufacturing to pharmaceutical development. The molar mass of KCl can serve as an exemplary case for understanding the chemistry of ionic compounds.
Key insights
Key Insights
- The molar mass of KCl serves as a foundational example in learning the chemistry of ionic compounds.
- Understanding atomic masses from the periodic table is essential for precise calculations.
- Grasping molar mass concepts is crucial for practical applications in the pharmaceutical and industrial sectors.
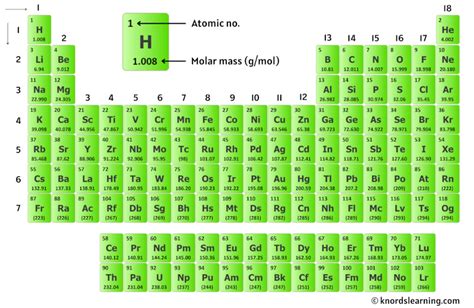
Exploring the Chemistry Behind KCl’s Molar Mass The molar mass of a compound like KCl, which consists of potassium (K) and chlorine (Cl), can be determined by summing the atomic masses of its constituent elements. Potassium has an atomic mass of approximately 39.10 g/mol, while chlorine has an atomic mass of roughly 35.45 g/mol. Adding these together, we find that the molar mass of KCl is approximately 74.55 g/mol. This straightforward calculation underscores the importance of knowing the precise atomic masses of elements from the periodic table, which is critical in any detailed chemical analysis.
Understanding KCl’s Structural and Chemical Properties The structure of KCl is quintessential in its simple ionic bond formation, with potassium donating an electron to chlorine, creating stable ions. The resulting ionic lattice structure imparts specific physical properties, such as high melting and boiling points and solubility in water. The molar mass calculation is not merely a theoretical exercise; it informs the understanding of these properties and guides predictions about the compound’s behavior in various environments, including biological systems.
FAQ section
What is the significance of knowing the molar mass of KCl in practical applications?
Knowing the molar mass of KCl is crucial in practical applications like pharmaceutical formulations, where precise dosing is essential. In industrial settings, it aids in calculating the required quantities of raw materials for production processes, ensuring cost-effectiveness and efficiency.
How does the molar mass concept apply to other ionic compounds?
The concept of molar mass applies universally to all compounds, whether ionic or covalent. For ionic compounds, it involves summing the atomic masses of all constituent ions, while for covalent compounds, it requires summing the atomic masses of all atoms in the molecule. This basic principle underpins quantitative chemistry and is essential for accurate chemical analysis and synthesis.
In conclusion, the molar mass of KCl is a fundamental aspect of chemical education that highlights the interplay between atomic masses and compound properties. It demonstrates the practical relevance of basic chemistry principles in both scientific research and industrial applications. Mastery of this concept paves the way for deeper understanding and expertise in the broader field of chemistry.