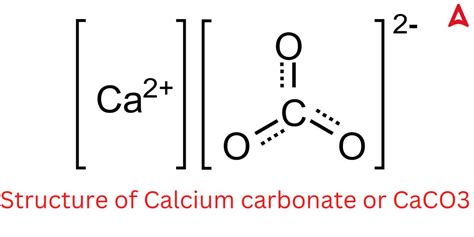
The molar mass of calcium carbonate, a seemingly simple concept, is often the starting point for understanding more complex chemical compositions. This compound, typically denoted as CaCO₃, is ubiquitous in nature, found in rocks, shells, and even human anatomy. Calculating the molar mass isn’t just an academic exercise but has practical implications in various fields, from pharmaceuticals to construction. Here, we delve into the intricacies of determining the molar mass of calcium carbonate and uncover some fascinating insights along the way.
The Basics of Molar Mass Calculation
To begin, molar mass refers to the mass of a given substance (typically in grams) per mole of the substance. The calculation is straightforward for compounds like calcium carbonate: it involves summing the atomic masses of all constituent atoms. Calcium (Ca), carbon ©, and oxygen (O) are the elements present in calcium carbonate. Using periodic table data, the atomic masses are:
- Calcium (Ca): approximately 40.08 g/mol
- Carbon ©: approximately 12.01 g/mol
- Oxygen (O): approximately 16.00 g/mol
Multiply the atomic masses by the number of atoms of each element in the compound and add them together:
40.08 (Ca) + 12.01 © + 3(16.00) (O) = 100.09 g/mol
Why This Matters
Knowing the molar mass is crucial in stoichiometry, the mathematical backbone of chemical reactions. It enables chemists to determine the relationships between reactants and products and quantify the amount of each substance needed for a given reaction. For instance, if you’re scaling up the production of calcium carbonate, understanding its molar mass is essential to accurately measure ingredients.
Key Insights
- Calculating the molar mass is foundational for chemical calculations.
- Accuracy in molar mass calculation ensures precise stoichiometric ratios in chemical reactions.
- Knowing the molar mass helps in scaling laboratory experiments to industrial production.
Applications in Real-World Scenarios
In the pharmaceutical industry, calcium carbonate is often used as a calcium supplement. Accurate dosing requires precise knowledge of its molar mass. An incorrect dosage can result in suboptimal or harmful effects for patients.
In the construction industry, calcium carbonate forms the basis for concrete and limestone, materials essential for building structures. Knowing its molar mass allows engineers to determine the precise mixture ratios of materials needed to achieve desired strength and durability in concrete.
The Relevance of Molar Mass Beyond Chemistry
Even in fields outside the immediate scope of chemistry, molar mass plays a significant role. For example, in environmental science, calcium carbonate’s role in carbon sequestration processes is of great interest. Accurate molar mass calculations help in understanding and quantifying the compound’s role in mitigating climate change by capturing atmospheric carbon dioxide.
How do you ensure the accuracy of molar mass calculations?
Accuracy in molar mass calculations is ensured by using the most up-to-date atomic masses from reliable sources such as the periodic table and double-checking the calculations for any arithmetic errors. Regular calibration of instruments used in chemical analysis also contributes to accuracy.
What happens if molar mass isn’t calculated correctly?
Miscalculations of molar mass can lead to significant errors in stoichiometry, resulting in incorrect proportions of reactants in a chemical reaction. This can alter the outcomes of experiments, affect the efficacy of products in industrial and pharmaceutical applications, and potentially compromise safety and efficiency.
Understanding the molar mass of calcium carbonate is essential not just for theoretical purposes but for a wide array of practical applications. From healthcare to construction and environmental science, this fundamental concept underpins a myriad of processes and innovations. With precise calculations, industries can optimize their operations, ensuring both efficacy and safety.