The quest to understand the molar mass of octane is a journey into the realm of chemistry that intersects with everyday life. Octane, a crucial component in gasoline, plays a significant role in the efficiency and performance of internal combustion engines. However, determining its molar mass isn’t merely an academic exercise—it’s essential for chemical calculations, lab work, and even environmental science. This guide will walk you through the practical steps and actionable advice to master the molar mass of octane, making sure to address common pain points and provide real-world examples to bolster your understanding.
The molar mass of a substance is the mass in grams of one mole of that substance. Understanding the molar mass of octane, a hydrocarbon with the molecular formula C8H18, can be crucial for chemists, engineers, and anyone involved in the study or application of fuels. Octane is often discussed in the context of its isomers, such as isooctane, which are evaluated for their octane numbers in gasoline. Calculating the molar mass of octane is foundational for these discussions, and knowing how to do it will empower you to tackle more complex chemical problems.
Quick Reference
Quick Reference
- Immediate action item: Write down the molecular formula of octane, C8H18.
- Essential tip: Break down the formula into its atomic components and use periodic table values to calculate each part.
- Common mistake to avoid: Don’t forget to multiply the atomic mass of each element by its occurrence in the formula.
By quickly familiarizing yourself with these key points, you can jump-start your understanding of how to approach the calculation of molar mass for octane.
Calculating Molar Mass of Octane: Step-by-Step
Calculating the molar mass of octane begins with a solid grasp of its molecular formula, C8H18. Here’s a detailed guide that will break down each component and explain the process:
Step 1: Understanding the Molecular Formula
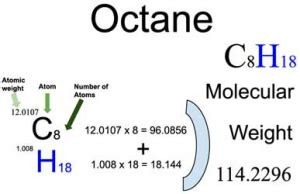
Octane has the molecular formula C8H18. This means that each molecule of octane is composed of 8 carbon © atoms and 18 hydrogen (H) atoms. Understanding this formula is the first critical step.
Step 2: Find the Atomic Masses
To find the molar mass, we need to look up the atomic masses of carbon and hydrogen on the periodic table:
| Element | Atomic Mass (g/mol) |
|---|---|
| Carbon © | 12.01 |
| Hydrogen (H) | 1.01 |
Step 3: Calculate the Total Mass for Carbon
Next, we calculate the total mass contributed by the carbon atoms in the molecule:
Number of carbon atoms × atomic mass of carbon = 8 × 12.01 g/mol = 96.08 g/mol
Step 4: Calculate the Total Mass for Hydrogen
Now, we do the same for the hydrogen atoms:
Number of hydrogen atoms × atomic mass of hydrogen = 18 × 1.01 g/mol = 18.18 g/mol
Step 5: Add the Masses Together
Finally, we sum the masses of the carbon and hydrogen atoms to get the molar mass of octane:
Molar mass of octane = 96.08 g/mol + 18.18 g/mol = 114.26 g/mol
This straightforward calculation provides us with the molar mass of octane, a fundamental piece of information for any chemistry-related task.
Advanced Calculations and Considerations
Once you’ve mastered the basic calculation, it’s useful to explore more advanced applications and considerations:
Calculations with Isomers
Octane has several isomers, such as n-octane and isooctane, each with slightly different molecular structures but the same molecular formula C8H18. The molar mass remains the same regardless of the isomer since the number of carbon and hydrogen atoms does not change. However, understanding isomers can be important in practical applications like fuel efficiency and performance.
Density and Molar Mass Relationship
The density of octane is approximately 0.703 g/mL. Knowing the molar mass allows us to calculate how much octane occupies a given volume, which is crucial in various scientific and engineering calculations.
For example, to find the number of moles in a given volume, we use the formula:
Number of moles = Volume (L) × Density (g/mL) / Molar Mass (g/mol)
Substituting the values:
Number of moles = 1 L × 0.703 g/mL / 114.26 g/mol ≈ 0.00614 moles
Practical FAQ
What are the common mistakes people make when calculating molar mass?
A common mistake is incorrectly identifying the atomic masses of elements or forgetting to multiply the atomic mass by the number of times an element appears in the molecule. For octane, a frequent error is miscounting the number of hydrogen atoms (18) or incorrectly applying the atomic mass of carbon (12.01 g/mol) and hydrogen (1.01 g/mol).
How does molar mass affect the use of octane in fuels?
Molar mass is integral when determining the precise amount of octane needed in fuel mixtures. For example, in the formulation of gasoline, knowing the molar mass ensures that the correct proportions of octane and other components are mixed to achieve the desired octane number, which affects engine performance and efficiency.
What are the implications of octane’s molar mass in environmental science?
In environmental science, understanding the molar mass of octane can help in modeling emissions and the combustion processes of fuels. Accurate calculations allow scientists to predict the behavior of pollutants released from engines and to devise strategies for mitigating their impact on air quality.
By understanding the molar mass of octane, you can navigate a variety of practical scenarios where precise chemical knowledge is essential, from engine performance tuning to environmental impact assessments.
In conclusion, the molar mass of octane, calculated as 114.26 g/mol, serves as a cornerstone for numerous chemical applications. Whether you’re delving into laboratory experiments, analyzing fuel compositions, or exploring environmental impacts, a firm grasp of this concept will enhance your scientific acumen and practical problem-solving abilities.