The molar mass of magnesium sulfate (MgSO4) is a fundamental concept in chemistry, one that holds substantial importance for understanding various chemical reactions, chemical formulations, and even biological processes. Knowing how to accurately determine the molar mass of MgSO4 is critical, whether you’re a student diving into organic or inorganic chemistry or a professional working in a laboratory. Let’s unravel the secrets behind this calculation, exploring not just the methodology but also its broader implications in scientific fields.
Key insights box:
Key Insights
- Accurately determining the molar mass of MgSO4 helps in chemical reactions and formulations.
- Each component’s atomic mass must be summed precisely for a correct calculation.
- Knowledge of molar mass is crucial for dosing and preparation in medical and pharmaceutical applications.
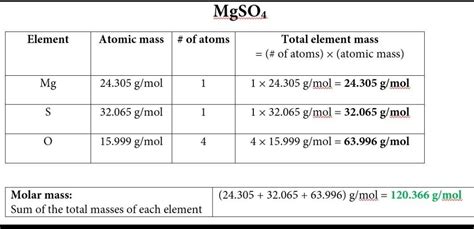
Determining the molar mass of MgSO4 involves a straightforward but precise calculation, combining the atomic masses of its constituent elements: magnesium (Mg), sulfur (S), and oxygen (O). The compound is composed of one magnesium atom, one sulfur atom, and four oxygen atoms. The atomic masses of these elements, according to the periodic table, are approximately 24.305 g/mol for magnesium, 32.06 g/mol for sulfur, and 16.00 g/mol for oxygen. To find the molar mass of MgSO4, simply add these atomic masses together as follows:
Mg (24.305 g/mol) + S (32.06 g/mol) + 4 × O (4 × 16.00 g/mol) = 24.305 + 32.06 + 64.00 = 120.365 g/mol
The Importance of Precision in Chemistry
In scientific research and industrial applications, precision is paramount. Small errors in molar mass calculations can lead to significant discrepancies in experimental outcomes. This is especially critical in drug formulation, where accurate dosages are necessary to ensure efficacy and safety. Understanding and applying the correct molar mass for MgSO4, or any other compound, ensures that chemists can design and conduct experiments with confidence, knowing that the foundational calculations are sound.
Real-World Application and Implications
Beyond the lab, the accurate determination of the molar mass of compounds like MgSO4 has profound real-world applications. In environmental science, precise calculations are vital for assessing pollutant concentrations and understanding the behavior of various chemicals in ecosystems. Additionally, in agriculture, accurate formulations of fertilizers and pesticides directly depend on precise chemical compositions, derived from accurate molar mass calculations. Hence, mastering the concept of molar mass extends beyond academic exercises into practical, impactful domains.
How does the molar mass of MgSO4 relate to its solubility in water?
The molar mass itself does not directly determine solubility; however, understanding the compound's properties aids in predicting its behavior in solution. MgSO4 is highly soluble in water due to its ionic nature, which is a property independent of its molar mass calculation but influenced by the strength of ionic bonds.
Why is it important to keep accurate records of molar mass calculations?
Accurate records of molar mass calculations ensure reproducibility and reliability in research and industrial settings. Errors in calculations can lead to incorrect formulations, potentially resulting in failures in experiments or adverse outcomes in manufacturing processes.
Understanding and accurately calculating the molar mass of compounds like MgSO4 is an essential skill in the scientific and medical fields. Precision in such calculations underpins the reliability and success of various applications, from laboratory experiments to real-world scenarios impacting health and the environment.