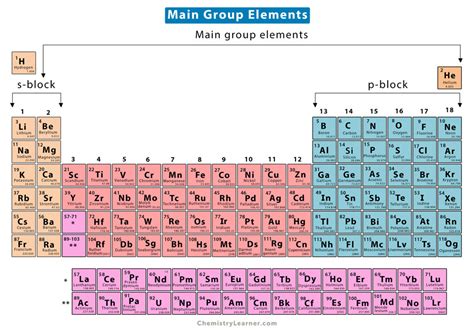
Understanding the chemistry of the main group elements is essential for anyone delving into the world of chemistry. These elements—comprising Groups 1, 2, 13-18 of the periodic table—play pivotal roles in numerous chemical reactions and everyday life phenomena. This guide aims to demystify the chemistry of these elements, offering step-by-step guidance, practical solutions, and actionable advice for both beginners and seasoned chemists.
It's common to feel overwhelmed when first approaching the complexities of main group elements chemistry. From grasping atomic structure to mastering their chemical reactivity, it can seem like a daunting task. However, this guide is designed to simplify these concepts into digestible segments, providing actionable steps, practical examples, and real-world applications that you can readily implement in your study and work. Whether you're a student looking to improve your understanding or a professional seeking a refresher, this guide will help you master the chemistry of main group elements effectively.
Immediate Steps to Understand Main Group Elements
To begin, focus on these immediate actions that will set you on the right path:
- Start with the basics: Ensure you have a solid understanding of atomic structure, electron configuration, and the periodic table.
- Use resources: Utilize online tutorials, videos, and textbooks to visualize concepts like electronegativity and ionization energy.
- Practice, practice, practice: Regularly work through example problems and chemical reactions involving main group elements to reinforce your knowledge.
Quick Reference: Main Group Elements Essentials
Quick Reference
- Immediate action item with clear benefit: Always start with electron configuration to predict reactivity.
- Essential tip with step-by-step guidance: When determining bond type, compare electronegativities.
- Common mistake to avoid with solution: Misunderstanding the significance of group trends in the periodic table.
Deep Dive into Alkali and Alkaline Earth Metals
The alkali metals (Group 1) and alkaline earth metals (Group 2) are known for their high reactivity, especially with water. Understanding their properties, reactivity trends, and common uses can significantly enhance your grasp of main group chemistry.
Alkali Metals: Lithium to Cesium
Alkali metals have a single electron in their outermost shell, making them highly reactive. This section covers their behavior in various chemical reactions, physical properties, and real-world applications.
- Example: The reaction of sodium (Na) with water (H2O):
2Na (s) + 2H2O (l) → 2NaOH (aq) + H2 (g)
This exothermic reaction demonstrates their high reactivity, producing sodium hydroxide and hydrogen gas.
Alkaline Earth Metals: Beryllium to Barium
Alkaline earth metals also have two electrons in their outermost shell. They are less reactive than alkali metals but still exhibit interesting chemical properties. This section explores their reactivity patterns, especially with acids and halogens, and their importance in industry.
- Example: Calcium carbonate (CaCO3) reacts with hydrochloric acid (HCl):
CaCO3 (s) + 2HCl (aq) → CaCl2 (aq) + CO2 (g) + H2O (l)
This reaction is fundamental in understanding acid-base interactions and is pivotal in processes like cement production.
Understanding Group 13 to 18 Elements
Elements in Groups 13 through 18, also known as post-transition metals, metalloids, and nonmetals, exhibit a diverse range of chemical behaviors. This section delves into the unique characteristics and chemical reactions of these elements.
Boron Group Elements (Group 13)
Boron and its neighbors (aluminum, gallium, indium, thallium) form a diverse set of compounds. They often function as semiconductors due to their intermediate electronegativity, making them crucial in electronics.
- Example: The reaction of boron trioxide (B2O3) with hydrogen fluoride (HF):
B2O3 (s) + 6HF (aq) → 2BF3 (g) + 3H2O (l)
This reaction is significant in the production of boron trifluoride, a key reagent in chemical synthesis.
Nonmetals and Noble Gases (Groups 15-18)
Nonmetals like nitrogen, oxygen, and the noble gases exhibit unique chemical inertness. Despite their varied properties, understanding their reactivity trends helps in grasping the fundamental principles of main group chemistry.
- Example: The formation of nitrogen dioxide (NO2):
2NO (g) + O2 (g) → 2NO2 (g)
This reaction is essential in understanding atmospheric chemistry, particularly in smog formation.
Practical FAQ: Navigating Main Group Elements Chemistry
Why do main group elements form compounds so differently?
Main group elements form compounds differently due to varying numbers of valence electrons and different electronic configurations. Alkali and alkaline earth metals, for instance, lose electrons easily to achieve a stable configuration, leading to ionic bonds. In contrast, nonmetals often share electrons to complete their octet, forming covalent bonds. Understanding these differences hinges on knowledge of their electron configurations and the periodic trends governing reactivity.
Best Practices for Studying Main Group Elements
Effectively studying main group elements chemistry involves several best practices:
- Use a periodic table as a reference for trends and properties.
- Relate chemical properties to electronic configurations for deeper understanding.
- Engage with interactive simulations and visualizations available online.
- Practice by solving diverse problem sets covering various reaction types and elements.
This guide serves as a foundational framework for mastering the chemistry of main group elements. By following these steps, tips, and practical examples, you’ll build a robust understanding of these essential elements, paving the way for advanced study and application in chemistry.