Understanding the NH3 (ammonia) Lewis structure is crucial for anyone studying chemistry. It's a foundation for grasping the basics of chemical bonding, molecular geometry, and even the behavior of gases. However, deciphering the NH3 Lewis structure can be daunting for beginners, especially when dealing with lone pairs, bonding pairs, and the distribution of electrons. This guide will break down the process into manageable steps, offering actionable advice, real-world examples, and solutions to common problems you might face.
Problem-Solution Opening: Conquering NH3 Lewis Structure Confusion
Many students feel overwhelmed when they first encounter the NH3 Lewis structure due to the complexity of electron distribution. The challenge lies in correctly placing lone pairs and bonding pairs while ensuring adherence to the octet rule. This guide aims to demystify this process, providing a step-by-step walkthrough that's both practical and professional. Whether you're prepping for an exam, conducting research, or simply trying to understand the basics better, this guide will equip you with the knowledge and confidence to master the NH3 Lewis structure efficiently.
Quick Reference
- Immediate action item with clear benefit: Start by sketching a simple skeleton structure of NH3 with nitrogen at the center and three hydrogen atoms surrounding it.
- Essential tip with step-by-step guidance: To fulfill the octet rule for nitrogen and hydrogen, pair electrons correctly and consider all available valence electrons.
- Common mistake to avoid with solution: Don't forget to count lone pairs on the nitrogen atom; it's easy to miss, but they're crucial for completing the Lewis structure.
How to Draw the NH3 Lewis Structure: Step-by-Step
Drawing the NH3 Lewis structure might seem challenging, but breaking it down into simpler steps can make it much more manageable. Follow this comprehensive guide to ensure you get it right the first time.
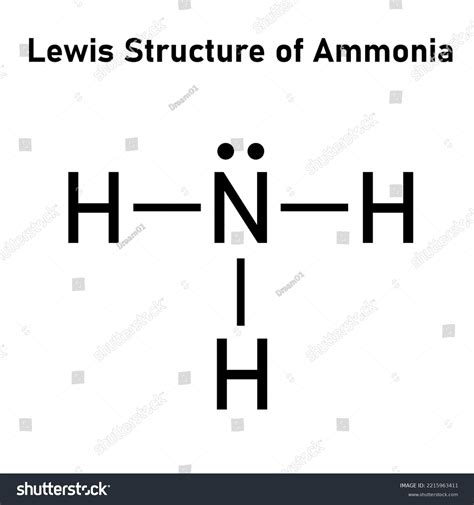
Step 1: Count Total Valence Electrons
First, you need to know how many valence electrons are available for the NH3 molecule. Nitrogen (N) is in group 15 of the periodic table and has 5 valence electrons. Each hydrogen (H) atom has 1 valence electron. Since there are three hydrogen atoms, you multiply 3 by 1, resulting in 3 valence electrons from hydrogen. Add these together:
5 (from N) + 3 (from H) = 8 valence electrons
Step 2: Determine the Central Atom
The central atom in NH3 is nitrogen. Less electronegative elements typically become the central atom in a molecule.
Step 3: Sketch a Simple Skeleton Structure
Place the nitrogen in the center with three hydrogen atoms bonded to it. This is a preliminary step to visualize the structure.
H
|
H - N - H
Step 4: Place Lone Pairs on the Central Atom
Nitrogen has 5 valence electrons, and it forms three single bonds with hydrogen, using up 6 electrons. To complete the octet rule, nitrogen still needs 2 electrons, which come in the form of a lone pair. Place one lone pair on the nitrogen atom.
Step 5: Check Electron Placement
Review the placement of your electrons. Hydrogen atoms should all have 2 electrons (forming a single bond), and nitrogen should have 8 electrons (3 bonding pairs and 1 lone pair).
Step 6: Confirm Octet Rule Compliance
Ensure that all atoms in the structure fulfill the octet rule. Hydrogen, being in group 1, only needs two electrons. Nitrogen, as a second-period element, follows the octet rule with eight electrons.
Step 7: Formal Charges
Calculate the formal charge on each atom. The formal charge equation is: (Valence electrons - Non-bonding electrons - 1/2 Bonding electrons) For nitrogen: 5 - 4 - 1/2(6) = 0 For hydrogen: 1 - 0 - 1/2(2) = 0 All atoms have a formal charge of 0, confirming the Lewis structure's correctness.
Practical FAQ: Frequently Asked Questions
Why is the NH3 molecule trigonal pyramidal?
The NH3 molecule adopts a trigonal pyramidal shape due to the presence of one lone pair on the nitrogen atom and the need to minimize electron-electron repulsion. The three hydrogen atoms and the lone pair on nitrogen occupy the vertices of a tetrahedron, but the lone pair takes up more space due to its larger electron cloud. This causes a distortion, resulting in a trigonal pyramidal shape.
Advanced Tips and Best Practices
Once you’re comfortable with the basics of the NH3 Lewis structure, consider these advanced tips to deepen your understanding:
- Electronegativity: Remember that nitrogen is more electronegative than hydrogen. This means that the shared electron pairs in the N-H bonds will be closer to the nitrogen atom.
- Resonance Structures: Although NH3 does not have resonance structures, understanding this concept can be useful for more complex molecules.
- Molecular Geometry Software: Utilize molecular geometry software to visualize NH3's structure, providing a three-dimensional understanding of its shape.
By following this guide, you’ll not only master the NH3 Lewis structure but also gain a solid foundation for tackling more complex molecules in chemistry.