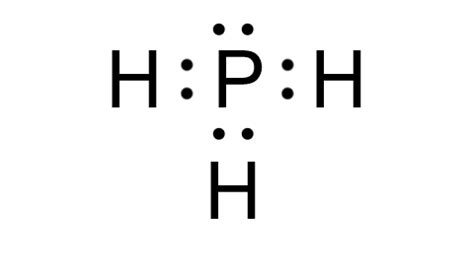
Understanding the Lewis Dot Structure of phosphorus trihydride (PH₃) is fundamental for anyone studying chemistry, as it aids in visualizing the bonding and lone pairs of electrons in the molecule. This guide will offer you a clear, step-by-step approach to mastering the Lewis Dot Structure of PH₃, complete with practical examples and expert tips. Let’s dive right in!
Introduction: The Problem-Solution Opening
Understanding molecular structures is pivotal for grasping chemical properties and reactivity. For many chemistry students and professionals, the Lewis Dot Structure of PH₃ can be a daunting challenge. PH₃, or phosphorus trihydride, presents unique challenges due to its unusual bonding characteristics compared to more familiar molecules like water or methane. However, with systematic approach and clear, actionable guidance, deciphering its Lewis Dot Structure becomes straightforward. This guide will arm you with the necessary tools and understanding to confidently tackle PH₃’s Lewis Dot Structure, ensuring that you can apply this knowledge to other similar molecules.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Draw a skeleton structure of PH₃ with phosphorus in the center and connect three hydrogen atoms.
- Essential tip with step-by-step guidance: Calculate the total number of valence electrons for all atoms involved, and distribute them following the octet rule and lone pairs.
- Common mistake to avoid with solution: Miscounting valence electrons or incorrectly placing lone pairs can lead to an incorrect Lewis Dot Structure. Always double-check your electron count and bonding arrangements.
Step-by-Step Guide to Drawing PH₃’s Lewis Dot Structure
To start drawing the Lewis Dot Structure of PH₃, you need to follow a series of methodical steps:
- Identify Valence Electrons: The first step is to calculate the total number of valence electrons. Phosphorus has 5 valence electrons, and each hydrogen has 1, meaning there are 3 hydrogen atoms in total, contributing 3 electrons. So, the total number of valence electrons in PH₃ is 8 (5 from phosphorus + 3 from hydrogen).
- Skeleton Structure: Place the phosphorus atom in the center, with three hydrogen atoms surrounding it in a trigonal pyramidal arrangement. This structure helps in visualizing the connectivity of the atoms. The hydrogen atoms are connected to phosphorus by single bonds.
- Distribute Electrons: Distribute the valence electrons starting with the bonds. Each single bond uses 2 electrons, and since there are three P-H bonds, this uses up 6 electrons. This leaves us with 2 electrons to place.
- Place Remaining Electrons: The remaining 2 electrons should be placed on the phosphorus atom to fulfill its octet. Phosphorus, being in group 15, can exceed the octet rule due to its ability to utilize its d-orbitals, leading to a total of 8 electrons around it.
This leaves us with the Lewis Dot Structure of PH₃. To review, phosphorus is surrounded by three hydrogen atoms, and the structure contains a lone pair on the phosphorus atom. This structure accurately represents the electron distribution and bonding in PH₃.
Understanding Electron Configuration
Understanding the electron configuration of PH₃ is crucial for comprehending its Lewis Dot Structure. Phosphorus, being in Group 15, has 5 valence electrons. Hydrogen, being in Group 1, has 1 valence electron. When forming PH₃, each hydrogen forms a single bond with the phosphorus atom. The bonding and lone pairs play a critical role in determining the molecule’s geometry and reactivity.
Why the Octet Rule Matters
The Octet Rule is a fundamental principle in chemistry which states that atoms tend to bond in such a way that each atom has eight electrons in its valence shell, providing stability. However, phosphorus can deviate from this rule when it has an expanded octet due to its ability to utilize its d-orbitals. In PH₃, phosphorus forms three covalent bonds with three hydrogen atoms and has a lone pair, contributing to its chemical stability despite not following the strict octet rule.
Common Mistakes to Avoid
To ensure the accuracy of your Lewis Dot Structure for PH₃, be mindful of these common pitfalls:
- Incorrect Electron Count: Double-check your total number of valence electrons. For PH₃, it’s critical to correctly sum the valence electrons from phosphorus and hydrogen atoms.
- Incorrect Bonding Arrangement: Ensure that the bonding configuration matches the expected geometry of the molecule (trigonal pyramidal for PH₃).
- Misplaced Lone Pairs: Ensure that lone pairs are placed on the central atom to satisfy its need for a complete valence shell, following the octet rule as closely as possible.
Practical FAQ
Why does PH₃ have a trigonal pyramidal shape?
PH₃ has a trigonal pyramidal shape because of the three single bonds from phosphorus to each hydrogen atom and the lone pair on the phosphorus atom. According to VSEPR (Valence Shell Electron Pair Repulsion) theory, these electron pairs repel each other, arranging themselves to maximize the distance between them, resulting in a pyramidal structure.
How does PH₃’s molecular geometry influence its properties?
The molecular geometry of PH₃ affects its properties significantly. The trigonal pyramidal shape and lone pair on phosphorus result in a polar molecule due to the uneven distribution of electron density. This polarity makes PH₃ a relatively strong base and allows it to participate in hydrogen bonding with other polar molecules.
Can PH₃ form more bonds despite having a lone pair?
PH₃, as it currently stands, does not have any additional valence electrons or empty orbitals available to form more bonds. The lone pair on phosphorus is sufficient to complete its valence shell, fulfilling the octet rule or allowing for an expanded octet. Any additional bonds would require an electron from one of the hydrogen atoms, which is not typical or energetically favorable for PH₃.
Conclusion
Grasping the Lewis Dot Structure of PH₃ empowers you with a deeper understanding of its chemical behavior and reactivity. This guide has walked you through each step to ensure a thorough understanding, avoiding common mistakes, and applying practical knowledge. By mastering this structure, you’re better prepared to tackle more complex molecules and expand your chemistry knowledge.