Mastering the Lewis Dot Structure for H2O: An Essential Guide
When it comes to understanding molecular structures, the Lewis dot structure provides a simplified yet profoundly useful representation. For water (H2O), mastering this concept is pivotal, whether you’re a chemistry student, a professional scientist, or someone who’s just curious about how molecules form. This guide is designed to take you through everything you need to know about the Lewis dot structure for H2O, providing clear and actionable advice that will make this complex topic easier to grasp.
Why Understanding H2O’s Lewis Dot Structure Matters
The Lewis dot structure for H2O is foundational for understanding how atoms bond together to form molecules. For water, it’s particularly significant because water is a molecule central to numerous chemical and biological processes. By mastering the Lewis dot structure for H2O, you’ll gain insight into bond formation, molecular shape, and reactivity, which are essential for advanced studies in chemistry and related fields. Moreover, understanding this concept helps demystify many real-world phenomena, from chemical reactions to the properties of water itself.Quick Reference
Quick Reference
- Immediate action item: Start by determining the total number of valence electrons for all atoms involved.
- Essential tip: Place the least electronegative atom in the center and arrange the more electronegative atoms around it.
- Common mistake to avoid: Forgetting to distribute lone pairs to satisfy the octet rule for all atoms.
Step-by-Step Guide to Drawing the Lewis Dot Structure for H2O
Understanding the step-by-step process to draw the Lewis dot structure for H2O involves several methodical stages. Here’s a detailed walkthrough, starting with the fundamentals.Step 1: Calculate Total Valence Electrons
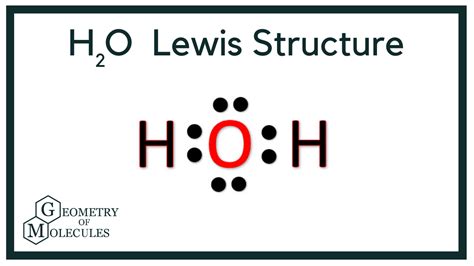
To start, determine the total number of valence electrons for the molecule. In the case of H2O:Hydrogen (H) has 1 valence electron, and Oxygen (O) has 6 valence electrons. Since there are two hydrogen atoms and one oxygen atom, the total number of valence electrons is:
2 (H) x 1 + 1 (O) x 6 = 2 + 6 = 8 valence electrons
Step 2: Identify the Central Atom
The central atom is typically the least electronegative element in the molecule, which for H2O is oxygen. Place it at the center:Central Atom: O
Step 3: Arrange the Hydrogen Atoms Around Oxygen
Hydrogen atoms will bond with the central oxygen atom. Since hydrogen only needs 2 electrons to complete its valence shell, it will bond with oxygen. Place two hydrogen atoms around the oxygen:O | H - H
Step 4: Distribute the Remaining Electrons
After placing hydrogen atoms around the oxygen, distribute the remaining valence electrons. Remember that each hydrogen atom already has two electrons from its bond with oxygen, so the remaining electrons must go to oxygen.Distribute lone pairs to oxygen first:
O : H - H
Total electrons used: 4 electrons (2 pairs) for bonding + 4 electrons (2 pairs) as lone pairs on oxygen
Step 5: Verify the Octet Rule
Ensure that each atom follows the octet rule (except hydrogen, which only needs two electrons). Oxygen has eight electrons around it (four from the lone pairs and four from bonds), fulfilling the octet rule. Each hydrogen has two electrons (one from each bond), also satisfying their need for stability.Step 6: Check Formal Charges
Calculate the formal charges for each atom to ensure the Lewis structure is the most accurate. Formal charge is calculated using the formula:Formal Charge = (Valence electrons of atom) - (Non-bonding electrons + ½ x bonding electrons)
For Oxygen (O): 6 (valence electrons) - 8 (non-bonding + bonding electrons) = 0 For Hydrogen (H): 1 (valence electrons) - 2 (bonding electrons) = 0Advanced Tips and Best Practices
While the basic steps are straightforward, several best practices will ensure your understanding is thorough.Redrawing If Needed
If the initial distribution doesn’t satisfy the octet rule, consider rearranging electrons. For instance, in H2O, our initial structure already satisfies the octet rule, so no further adjustments are necessary.Recognizing Exceptions
Keep in mind that there are exceptions to the octet rule, particularly in molecules with expanded octets or with elements like boron or hydrogen. H2O is not an exception as it follows the standard octet rule.Utilizing Molecular Geometry
Understanding the VSEPR (Valence Shell Electron Pair Repulsion) theory helps predict the three-dimensional shape of molecules based on the electron pairs around the central atom. For H2O, the molecular geometry is bent or angular due to two lone pairs on the oxygen atom.FAQ Section
Why is the Lewis dot structure important?
The Lewis dot structure is crucial because it helps visualize the arrangement of atoms and electrons in a molecule, aiding in understanding how chemical bonds form, how molecules interact, and predicting molecular shapes and properties.
What does the lone pair of electrons on oxygen signify?
The lone pairs on oxygen indicate areas of electron density that do not participate in bonding. In H2O, the two lone pairs influence the molecule’s bent shape and explain why water has its unique properties, such as its polarity and ability to form hydrogen bonds.
How do formal charges affect Lewis structure selection?
Formal charges help determine the most stable and accurate Lewis structure. A structure with the smallest formal charges, ideally zero, is more likely to be correct. This is because minimizing the formal charge discrepancy indicates a closer match to the actual charge distribution in the molecule.
Final Thoughts
Mastering the Lewis dot structure for H2O is an excellent exercise in fundamental chemical principles. By carefully following the steps outlined in this guide, you’ll be able to accurately represent the molecular structure of water. Remember, practice makes perfect. Try drawing Lewis structures for different molecules to further hone your skills. Whether your interest lies in academic or professional chemistry, these foundational skills will serve you well in understanding and predicting chemical behaviors.Your journey into the molecular world has just begun. With this guide, you have all the tools needed to decode the simplicity and complexity of water’s Lewis dot structure. Happy learning!