Understanding CS2 Lewis Dot Structure is essential for anyone delving into the realm of chemical bonding and molecular geometry. This guide will walk you through the step-by-step process to grasp the concepts, using practical examples and tips to avoid common mistakes. By the end of this guide, you will have a solid understanding of how to draw and interpret CS2 Lewis Dot Structures.
Problem-Solution Opening Addressing User Needs (250+ words)
The realm of chemistry often presents challenging topics, such as Lewis Dot Structures, which can seem daunting at first glance. CS2, or carbon disulfide, is one such molecule whose Lewis Dot Structure may appear complex due to its unique bonding. For many, the first encounter with this topic can lead to confusion and frustration. The purpose of this guide is to demystify the Lewis Dot Structure of CS2 by providing a step-by-step, practical, and problem-solving approach. This guide aims to empower you with the knowledge and confidence to not only draw the Lewis Dot Structure for CS2 but to understand its geometry and bonding. We will focus on making this process as straightforward as possible, leveraging real-world examples and avoiding common pitfalls. By the end of this guide, you'll see that mastering the CS2 Lewis Dot Structure is within your grasp.
Quick Reference
Quick Reference
- Immediate action item: Count the total valence electrons for CS2 to start the structure drawing.
- Essential tip: Place carbon in the center and surround it with two sulfur atoms, following the octet rule.
- Common mistake to avoid: Failing to correctly distribute electrons can lead to an incorrect structure; double-check the placement and bonding.
Detailed How-To Sections
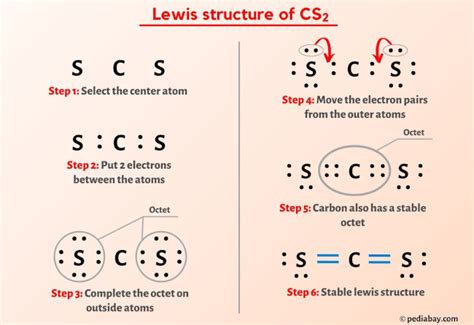
How to Count Valence Electrons for CS2
The first step in drawing a Lewis Dot Structure is to determine the total number of valence electrons. Carbon has four valence electrons, and each sulfur atom contributes six valence electrons. Therefore, for CS2:
Total valence electrons = 4 (Carbon) + 6 (Sulfur) x 2 = 16 valence electrons.
Here’s a breakdown of this calculation:
- Carbon ©: 4 valence electrons
- Sulfur (S): 6 valence electrons each, and there are two sulfur atoms.
This understanding sets the stage for accurately sketching the Lewis Dot Structure.
Drawing the Lewis Dot Structure
To draw the Lewis Dot Structure for CS2, follow these steps:
- Step 1: Place the central atom (carbon) in the middle and surround it with the two sulfur atoms.
- Step 2: Connect each sulfur atom to carbon with a single bond.
- Step 3: Distribute the remaining valence electrons to satisfy the octet rule for each atom.
Here’s a visual guide:
Carbon forms single bonds with each sulfur atom:
S
|
C - S
Next, distribute the remaining valence electrons:
Each sulfur needs three more electrons to complete its octet, and carbon will use its four valence electrons to form bonds:
..
|
C = S
|
S
Now, let’s place the extra electrons:
..
|
C = S
: S
Ensure that each sulfur atom has eight electrons and carbon has four bonded electrons. This ensures that the octet rule is satisfied for all atoms involved.
Understanding Electron Distribution and Bonding
A crucial aspect of drawing the Lewis Dot Structure is understanding how to distribute electrons correctly:
- Carbon shares its valence electrons to form single bonds with both sulfur atoms.
- Each sulfur atom needs three more electrons to complete its octet.
- Distribute the remaining electrons (from the total 16) so that each atom ends up with an octet.
Carbon, being the central atom, will use its valence electrons primarily to form bonds with the sulfur atoms. The lone pairs on sulfur will help complete their octets.
Practical FAQ
What if I find it hard to distribute the electrons correctly?
If distributing electrons seems overwhelming, follow these specific steps:
- Start by forming single bonds between the central atom and each surrounding atom.
- Use remaining electrons to fill the octets of surrounding atoms first.
- Distribute the rest to the central atom, if needed.
- Always check if every atom satisfies the octet rule.
This systematic approach can make electron distribution much easier.
Why is the CS2 molecule linear?
CS2 is a linear molecule because of the arrangement of its atoms and the way electrons are distributed. Carbon forms two double bonds with sulfur atoms, resulting in a linear geometry. The sulfur atoms are 180 degrees apart from each other along the same line as the carbon atom.
To visualize this, remember that carbon is sp hybridized in CS2, leading to a linear structure. This hybridization allows for the optimal arrangement of the atoms, minimizing electron repulsion.
How do I verify the correctness of my Lewis Dot Structure?
To verify the correctness of your Lewis Dot Structure, follow these steps:
- Check if the total number of electrons you started with is the same as the total number of electrons in your structure.
- Ensure that each atom satisfies the octet rule (except for hydrogen, which follows the duet rule).
- Look for formal charges and try to minimize them. The most stable structure will have the lowest formal charges.
By carefully checking these aspects, you can confirm the accuracy of your Lewis Dot Structure.
Advanced Tips and Best Practices
As you become more comfortable with drawing Lewis Dot Structures, consider these advanced tips:
- Practice drawing different molecules to get a feel for various bonding scenarios.
- Utilize molecular model kits to physically manipulate atoms and understand spatial arrangements.
- Review hybrid orbital theory to better understand the geometries of more complex molecules.
Keep in mind that becoming proficient with Lewis Dot Structures requires consistent practice and application to various molecules.
This guide has provided you with a comprehensive, step-by-step approach to mastering the Lewis Dot Structure for CS2. By following these instructions and utilizing the tips and best practices mentioned, you will gain a deeper understanding and confidence in drawing and interpreting molecular structures. Remember, practice makes perfect, and with time, you will find this process second nature.