Determining whether NH3 (ammonia) is an acid or a base is a fundamental question in chemistry that delves into the behavior of compounds in aqueous solutions. Many students and professionals find themselves puzzled about this, as ammonia doesn’t fit neatly into the classic definitions of acids and bases. In this comprehensive guide, we’ll walk you through the specifics with clear explanations, actionable advice, and practical examples to ensure you fully understand NH3's role as a chemical species. By the end, you'll be equipped with the knowledge to confidently address the question, "Is NH3 acid or base?" and much more.
Problem-Solution Opening Addressing User Needs
You might come across NH3 (ammonia) in a variety of contexts—laboratory settings, everyday life, and even environmental studies. Its chemical nature can seem ambiguous because it exhibits properties of both acids and bases. For instance, when NH3 interacts with water, it doesn’t always fit the conventional mold of how we understand acids and bases. This confusion often leads to challenges in grasping its role in different chemical reactions. In essence, NH3 is more accurately classified as a base because it can accept protons and donate electrons, but it’s essential to understand its behavior in detail for practical applications in various fields like environmental chemistry, biochemistry, and industrial processes.
In this guide, we provide a step-by-step breakdown of NH3’s nature, supplemented with actionable tips, real-world examples, and clear explanations. We’ll tackle this through a practical lens, offering you the insights and tools needed to comprehend NH3 without ambiguity.
Quick Reference
Quick Reference
- Immediate action item: Understand that NH3 is a base because it accepts protons and increases the pH of a solution.
- Essential tip: When working with NH3 in aqueous solutions, remember its role as a Bronsted-Lowry base—it donates a pair of electrons to hydrogen ions.
- Common mistake to avoid: Don’t assume that NH3 only behaves as a base; it also acts as a Lewis base by donating a pair of electrons.
Understanding NH3: Is it an Acid or a Base?
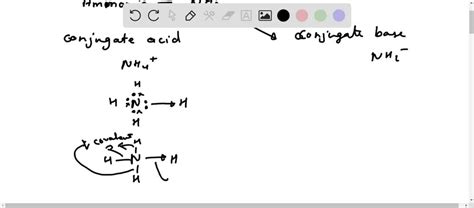
NH3 is fundamentally a base, but to understand why, we need to delve into the theories of acids and bases. The simplest classification is based on the Bronsted-Lowry theory, which defines acids as proton donors and bases as proton acceptors. When NH3 is added to water, it accepts a hydrogen ion (H+), forming NH4+ and OH-, thus increasing the pH of the solution.
Aside from the Bronsted-Lowry theory, there’s also the Lewis theory of acids and bases. In this context, acids are electron pair acceptors, and bases are electron pair donors. NH3 donates a lone pair of electrons to make it a Lewis base as well.
Here’s how it breaks down:
- Bronsted-Lowry Base: NH3 accepts H+ ions.
- Lewis Base: NH3 donates a pair of electrons.
Detailed How-To: Understanding Ammonia’s Role as a Base
To deeply understand why NH3 is classified as a base, we’ll break down its chemical behavior and properties through various dimensions.
Chemical Properties of NH3
NH3 has several defining chemical properties:
- Molecular structure: NH3 consists of one nitrogen atom bonded to three hydrogen atoms.
- Lone pair on nitrogen: Nitrogen has a lone pair of electrons that it can readily donate.
- Aqueous solution behavior: In water, NH3 accepts a proton from water (H2O), forming NH4+ and OH-, thus increasing the pH.
Bronsted-Lowry Theory and NH3
According to the Bronsted-Lowry theory, acids donate protons (H+) and bases accept protons:
When NH3 is introduced to a solution, it can accept H+ from water:
H2O + NH3 ↔ NH4+ + OH-
This reaction clearly shows NH3 as the base, accepting a proton to form NH4+, and producing OH- ions which elevate the pH.
Lewis Theory and NH3
The Lewis theory broadens the concept of acids and bases:
According to this theory, acids accept electron pairs, while bases donate electron pairs:
NH3 donates its lone pair of electrons to form coordinate covalent bonds with various acceptors:
NH3 + BF3 ↔ F2B-NH2
Here, NH3 donates its lone pair to BF3, making it a Lewis base.
Real-World Application of NH3 as a Base
Understanding NH3’s role as a base has numerous practical applications:
- Chemical manufacturing: NH3 is used to produce various industrial chemicals, often acting as a base in reactions.
- Biochemical reactions: In biological systems, NH3 helps in forming amino acids, where it plays the role of a base by accepting protons.
- Environmental chemistry: Understanding the behavior of NH3 in water helps in managing eutrophication, where NH3 is a major factor.
Practical Examples to Solidify Your Understanding
Let’s delve into practical examples that highlight NH3’s behavior as a base.
Example 1: NH3 in Laboratory Reactions
In laboratories, NH3 often participates in reactions to form salts:
NH3 + HCl → NH4Cl
In this reaction, NH3 accepts H+ from HCl, acting as a base.
Example 2: Soil pH Adjustment
NH3 is used to adjust soil pH because it’s a weak base:
When applied to acidic soils, NH3 neutralizes acidity:
NH3 + H+ → NH4+
This application is crucial for enhancing crop growth and soil health.
Example 3: Industrial Production
In industrial processes, NH3 is crucial for producing nitric acid:
Through the Ostwald process, NH3 is oxidized to produce nitric acid: