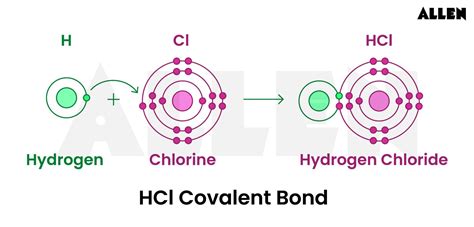
Have you ever found yourself puzzled by the difference between ionic and covalent bonds, especially when encountering complex compounds like HCl? This guide aims to clear up any confusion and provide you with the knowledge to confidently understand whether HCl is ionic or covalent. By the end of this guide, you’ll be equipped with actionable steps to tackle similar chemistry questions and will have a solid grasp of bonding types.
Understanding the Problem: Why Bonding Matters
At the heart of chemistry lies the concept of bonding, which determines the structure, properties, and reactivity of compounds. Ionic and covalent bonds represent the two main types of chemical bonds, and knowing the difference between them can significantly impact how we approach chemical reactions and compounds. When it comes to HCl, identifying whether it’s ionic or covalent is essential for grasping its behavior in various chemical contexts.
The dilemma often arises because some compounds can exhibit characteristics of both ionic and covalent bonding. Therefore, it's important to approach each compound with a systematic understanding of bonding principles rather than relying solely on memorization.
Quick Reference
Quick Reference
- Immediate action item: Determine the difference between ionic and covalent bonds using electronegativity values.
- Essential tip: Follow a step-by-step guide to analyze the bonding in HCl.
- Common mistake to avoid: Assuming HCl is ionic due to misconceptions about bond types.
Detailed How-To: Analyzing the Bonding in HCl
To determine whether HCl is ionic or covalent, it’s essential to understand the fundamental concepts and follow a systematic approach.
Step-by-Step Guide to Bonding Analysis
Follow these steps to distinguish between ionic and covalent bonds:
- Identify the elements: HCl consists of hydrogen (H) and chlorine (Cl).
- Examine electronegativity: Electronegativity refers to the ability of an atom to attract electrons in a chemical bond. A key factor in determining bond type is the difference in electronegativity between the bonded atoms.
- Compare electronegativity values: For hydrogen, the electronegativity value is 2.1, while for chlorine, it is 3.16. The difference between these values is 1.06.
When the electronegativity difference is greater than 1.7, a bond is typically considered ionic. In contrast, if the difference is less than 1.7, the bond is usually covalent.
Given the electronegativity difference of 1.06 between hydrogen and chlorine, we might think it’s a covalent bond. However, this scenario is more complex.
To understand why HCl is not purely ionic but has characteristics of both, we need to delve deeper:
- Partial Ionic Character: In reality, the bond between hydrogen and chlorine exhibits a degree of ionic character due to the difference in electronegativity, but it is not purely ionic.
- Polar Covalent Bond: The electronegativity difference results in a polar covalent bond where the shared electrons are pulled more towards the chlorine atom, leading to a partial negative charge on chlorine and a partial positive charge on hydrogen.
Practical Examples to Understand Better
Here are some practical examples to cement your understanding:
- In NaCl, the electronegativity difference is significantly higher (≈3.16), leading to a purely ionic bond.
- In contrast, in H2O, the electronegativity difference between hydrogen and oxygen (3.44) results in a polar covalent bond.
From these examples, you can see how electronegativity influences the type of bonding, and how HCl falls somewhere between purely ionic and covalent bonds.
Practical FAQ: Addressing Common Questions
Is HCl ionic or covalent?
HCl is not purely ionic or covalent but exhibits characteristics of both due to the partial ionic character in the polar covalent bond between hydrogen and chlorine.
How do you determine if a bond is ionic, covalent, or polar covalent?
To determine the type of bond, you should calculate the electronegativity difference between the bonded atoms. If the difference is greater than 1.7, it’s likely ionic. If it's between 0.4 and 1.7, it’s likely covalent. If it’s between 1.7 and 3.5, the bond is polar covalent, exhibiting characteristics of both ionic and covalent.
Why does HCl have a polar covalent bond?
HCl has a polar covalent bond because the electronegativity difference between hydrogen (2.1) and chlorine (3.16) results in a partial negative charge on chlorine and a partial positive charge on hydrogen. This uneven distribution of electron density leads to the bond's polarity.
By thoroughly understanding the concepts and applying them through systematic analysis, you can determine whether compounds like HCl are ionic, covalent, or polar covalent, which will significantly enhance your chemistry knowledge.
Tips and Best Practices
Here are some tips to ensure you grasp the concepts of ionic and covalent bonding fully:
- Use Electronegativity Values: Familiarize yourself with common electronegativity values for different elements to determine bond types effectively.
- Practice Calculations: Regularly practice calculating electronegativity differences to solidify your understanding.
- Consult Reference Materials: Use chemistry textbooks and online resources to consult detailed explanations and examples.
- Visualize the Bonds: Draw Lewis structures and visualize the electron distribution to better understand bonding types.
By integrating these tips into your study routine, you will develop a stronger, more intuitive grasp of chemical bonding, making it easier to tackle various chemistry questions confidently.
Remember, chemistry is both a science and an art. By combining systematic study, practical examples, and visualization, you can unveil the mysteries behind compounds like HCl, enhancing your overall chemistry proficiency.