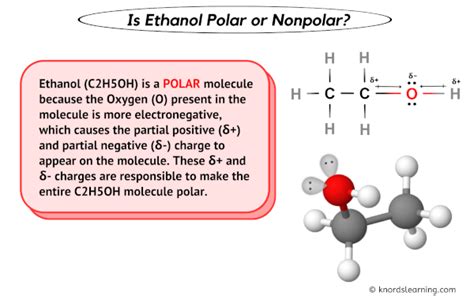
Understanding ethanol polarity involves delving into the molecular structure and the nature of its chemical bonds. Ethanol, a simple alcohol, holds significant relevance in various fields including organic chemistry, biochemistry, and even as a biofuel. Recognizing its polarity is critical for applications ranging from industrial solvents to metabolic pathways in biological systems.
In essence, ethanol is composed of an ethyl group (C2H5) and a hydroxyl group (OH). The hydroxyl group is the key to ethanol’s polarity. This functional group has an inherent dipole moment due to the difference in electronegativity between oxygen and hydrogen atoms. Oxygen’s higher electronegativity attracts electrons more strongly than hydrogen does, creating a partial negative charge (δ-) near the oxygen and a partial positive charge (δ+) around the hydrogen. This electronegativity difference facilitates the formation of hydrogen bonds and influences ethanol’s interactions with other polar and non-polar molecules.
Key Insights
- Ethanol’s polarity arises from the electronegativity difference in the hydroxyl group.
- Its polarity allows for effective hydrogen bonding, enhancing solubility in water.
- This property is exploitable for its use as a solvent in both organic reactions and biological contexts.
Structural Components of Ethanol
The structure of ethanol can be denoted as CH3CH2OH. The ethyl group, or alkanes, consists of non-polar carbon-hydrogen bonds. However, the molecule’s overall polarity stems from the polar nature of the hydroxyl group. The electronegativity difference between carbon, hydrogen, and oxygen creates regions of charge that lead to the molecule’s overall dipole moment, which is responsible for many of ethanol’s chemical behaviors.
Chemical Properties and Interactions
Ethanol's polarity profoundly influences its chemical properties and interactions. Its ability to form hydrogen bonds with other polar molecules, such as water, is a cornerstone of its utility. This characteristic enhances ethanol's solubility in water, making it an excellent solvent for many polar compounds. For instance, ethanol is widely used in the extraction and purification processes in organic chemistry due to its capacity to dissolve both polar and non-polar substances. Furthermore, the hydrogen bonds ethanol can form are vital in biological systems where ethanol interacts with proteins, nucleic acids, and other biomolecules, thereby influencing various cellular functions.
Can ethanol mix with non-polar solvents?
Yes, ethanol can mix with non-polar solvents due to its dual nature. While its hydroxyl group makes it polar, the ethyl group’s non-polar nature allows ethanol to be somewhat compatible with non-polar solvents, a property exploited in various organic synthesis reactions.
What is the role of ethanol in biological systems?
In biological systems, ethanol acts as an intermediary in metabolic processes. It is produced during fermentation and can be broken down by the liver. Its interaction with proteins and other molecules can affect cellular functions and enzyme activities, contributing to its role in both metabolic pathways and toxicological effects.
To summarize, ethanol’s polarity is a crucial factor determining its chemical behavior, solvent capabilities, and interactions within biological systems. By understanding the electronegativity differences and the resultant dipole moment, we gain valuable insights into its applications in various scientific and industrial fields.