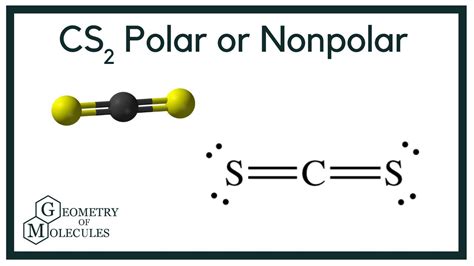
Carbon disulfide (CS2) is a fundamental chemical compound with profound implications in various industrial and academic fields. Despite its simplicity, understanding the polarity of CS2 unveils a wealth of information regarding its behavior in different environments. In this article, we delve into the specifics of CS2’s polarity, examining it from both a theoretical and practical standpoint, backed by evidence-based statements and real-world examples.
Key Insights
- CS2 has a linear structure which directly impacts its polarity.
- The symmetrical arrangement of sulfur atoms around the central carbon atom cancels out any dipole moments.
- This results in CS2 being a nonpolar molecule, crucial for understanding its solubility and reactivity.
Molecular Geometry and Polarity
The shape of a molecule plays a pivotal role in determining its polarity. For CS2, the molecule adopts a linear geometry with sulfur atoms on either side of the central carbon atom. According to molecular orbital theory, when atoms bond in such a symmetrical fashion, any resulting dipole moments tend to cancel each other out. This cancellation effect is crucial for understanding CS2’s nonpolar nature.The linear configuration of CS2 means that the dipole moment vectors generated by the carbon-sulfur bonds point in directly opposite directions. Hence, these vectors counterbalance each other, leading to an overall net dipole moment of zero. This fundamental insight helps explain CS2’s behavior in solvents and its reactions with other nonpolar substances.
Electronegativity and Bond Polarity
To comprehend CS2’s polarity, one must consider the concept of electronegativity, which is the ability of an atom to attract electrons towards itself in a chemical bond. In the case of CS2, the electronegativity difference between carbon and sulfur is relatively small. Sulfur, with an electronegativity of 2.58, and carbon, with 2.55, share electrons almost equally, resulting in a nonpolar covalent bond.The bond polarity between carbon and sulfur does not create significant charge separation. As a result, CS2 does not exhibit the behavior typical of polar molecules, such as forming hydrogen bonds or dissolving well in polar solvents like water. Instead, CS2 is more likely to interact with and dissolve in nonpolar solvents, which is a key practical insight for chemists working with this compound.
What implications does CS2’s nonpolarity have for its applications?
CS2’s nonpolar nature means it can be used as a solvent for nonpolar substances, including fats, resins, and certain polymers. This makes it valuable in industries such as organic synthesis, where nonpolar solvents are preferred for reactions involving nonpolar reactants.
Why is understanding the polarity of CS2 important?
Understanding the polarity of CS2 helps predict its interactions with other molecules and its environmental impact. For instance, CS2 is volatile and can form mists in the atmosphere, which can be harmful to health due to its toxicity, even though it is nonpolar. Thus, handling and disposal must be done with care.
In conclusion, the nonpolar nature of CS2 is a defining characteristic that influences its applications, interactions, and handling. This knowledge, grounded in molecular geometry and electronegativity, is indispensable for both theoretical studies and practical industrial uses. The exploration of CS2’s polarity underscores the intricate balance that governs molecular interactions, providing a deeper understanding of this important chemical compound.