Understanding ionic bonds is crucial for anyone diving into chemistry, whether you are a student, a professional in the field, or just someone curious about the molecules and compounds that make up the world around us. Ionic bonds form when there is a transfer of electrons between a metal and a non-metal atom, resulting in oppositely charged ions that attract each other. This guide will delve into real-world examples of ionic bonds, offering practical insights, tips, and troubleshooting advice to ensure you grasp the concept thoroughly. Let's break down this fundamental chemical concept, addressing common pain points and offering a clear pathway from basic to advanced understanding.
Understanding the Basics of Ionic Bonds
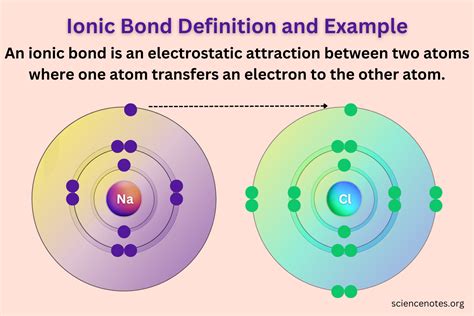
Ionic bonds are formed through the electrostatic attraction between positively charged cations and negatively charged anions. These bonds are essential for the structure and stability of many compounds we encounter daily. For instance, table salt (sodium chloride, NaCl) is a quintessential example of an ionic compound. Here’s a quick reference to get you started:
Quick Reference
- Immediate action item with clear benefit: Identify the elements in your compound to determine if they are metals or non-metals to check for the possibility of an ionic bond.
- Essential tip with step-by-step guidance: Always remember that ionic bonds are typically formed when a metal transfers electrons to a non-metal, resulting in cations and anions.
- Common mistake to avoid with solution: Confusing ionic bonds with covalent bonds; ensure you note the transfer of electrons rather than the sharing of electrons.
Detailed Exploration of Sodium Chloride
To grasp ionic bonds, let’s delve deeply into the most familiar example: sodium chloride (NaCl). Sodium (Na) is a metal from Group 1 of the periodic table, and chlorine (Cl) is a non-metal from Group 17. Sodium has one electron in its outermost shell, while chlorine has seven electrons in its outermost shell. When sodium loses its single outermost electron, it becomes a sodium cation (Na+), and chlorine, gaining that electron, becomes a chloride anion (Cl-).
Here’s a step-by-step breakdown of how sodium chloride forms:
- Electron Transfer: Sodium donates its single outer electron to chlorine.
- Formation of Ions: The result is Na+ (sodium cation) and Cl- (chloride anion).
- Attraction: The oppositely charged ions attract each other, forming an ionic bond.
- Crystal Lattice: These bonds arrange the ions in a crystal lattice structure, providing the solid, crystalline structure of table salt.
The formation of sodium chloride is not just a classroom example but is also found in natural deposits like the Dead Sea and the salt we use in our kitchens. The beauty of ionic bonding lies in its predictability and the ability to describe and predict the behavior and properties of ionic compounds.
Magnesium Oxide: A Deeper Dive
Next, let’s examine magnesium oxide (MgO) to understand another example of an ionic bond. Magnesium is a metal from Group 2, and oxygen is a non-metal from Group 16. Magnesium has two outer electrons that it can easily lose to achieve a stable electron configuration similar to that of a noble gas. Oxygen, with six electrons in its outer shell, needs two more to complete its octet.
Here’s a detailed look at how magnesium oxide forms:
- Electron Transfer: Magnesium loses its two outer electrons to become Mg2+.
- Formation of Ions: Oxygen gains two electrons to become O2-.
- Attraction: The Mg2+ and O2- ions attract each other, forming an ionic bond.
- Structure: The ionic bond creates a crystalline lattice structure that is highly stable and has a high melting point, characteristic of many ionic compounds.
Magnesium oxide serves various practical purposes. For example, it is used as a refractory material in high-temperature environments, such as in furnace linings, because of its high melting point. This compound illustrates how ionic bonds dictate the physical and chemical properties of materials we use in industrial applications.
Practical Examples in Everyday Life
Ionic bonds are not confined to laboratories; they play an essential role in many everyday products. Here are a few real-world examples:
- Table Salt (NaCl): We use it for seasoning and preserving food.
- Epsom Salt (MgSO4): Used in baths and for various health benefits.
- Aluminum Fluoride (AlF3): Found in dental products to help strengthen tooth enamel.
Each of these compounds forms through ionic bonding and exemplifies how this fundamental chemical concept underpins various aspects of our daily lives.
Addressing Common Challenges
Learning about ionic bonds can sometimes present hurdles, but here are some practical solutions to help you overcome them:
- Distinguishing Between Ionic and Covalent Bonds: Focus on the transfer of electrons (indicative of ionic bonds) versus the sharing of electrons (covalent bonds).
- Understanding Lattice Energy: Remember that lattice energy is the energy required to separate one mole of a solid ionic compound into its gaseous ions, reflecting the strength of ionic bonds.
- Predicting Solubility: Ionic compounds generally dissolve in polar solvents like water due to the attraction between the compound’s ions and the polar solvent molecules.
Practical FAQ
How can I tell if a compound is ionic or covalent?
To determine if a compound is ionic or covalent, examine the elements involved. If the compound includes a metal and a non-metal, it’s likely ionic. For example, NaCl (sodium chloride) is ionic because sodium (a metal) transfers an electron to chlorine (a non-metal). If you have two non-metals, the compound is likely covalent, such as H2O (water), where hydrogen and oxygen share electrons.
Why do ionic compounds have high melting points?
Ionic compounds have high melting points because of the strong electrostatic forces between the oppositely charged ions in their crystal lattice structure. These strong bonds require a substantial amount of energy to break, thus leading to high melting points. For example, magnesium oxide (MgO) has a melting point of about 2852°C due to the strong ionic bonds.
Can ionic compounds conduct electricity?
Ionic compounds do not conduct electricity in their solid state due to the fixed positions of the ions in the crystal lattice. However, they can conduct electricity when dissolved in water or in a molten state. This is because the free movement of ions in these states allows for the flow of electrical charge. For instance, when NaCl is dissolved in water, the Na+ and Cl- ions can move freely and conduct electricity.
This guide provides a robust framework for understanding ionic bonds through practical examples and step-by-step explanations. By tackling real-world examples and addressing common questions, you can gain a deeper appreciation and comprehension of ionic bonding, empowering you to apply this knowledge across various scientific fields.