Understanding the conversion from grams to moles is a fundamental skill in chemistry that can help you master various complex chemical concepts. Whether you’re a student or a professional, navigating this conversion process can be daunting if you lack the right guidance and practical examples. This guide is designed to walk you through each step, providing actionable advice, real-world examples, and tips to make your learning journey smooth and effective.
Introduction: Why Mastering Grams to Moles Conversion is Essential
Mastering the conversion from grams to moles is crucial for anyone involved in chemistry. This skill enables you to quantify substances in chemical reactions, understand stoichiometry, and predict the outcomes of chemical processes. By learning to convert grams to moles, you can better comprehend molecular relationships, reaction proportions, and chemical formulations, all of which are essential for academic success and professional proficiency in chemistry.
In this guide, we will break down the complex process of converting grams to moles into straightforward, manageable steps. You’ll find practical examples, actionable advice, and a clear understanding of the underlying concepts that make this process seamless.
Quick Reference Guide
Quick Reference
- Immediate action item: Start by identifying the molar mass of the substance from the periodic table.
- Essential tip: Use the formula moles = grams / molar mass to convert grams to moles.
- Common mistake to avoid: Ensure that your unit of grams is correctly converted to moles by ensuring the molar mass unit is in grams per mole.
Step-by-Step Conversion Process
Let’s start with the foundational steps required to convert grams to moles. Understanding these steps thoroughly will give you a robust foundation for future calculations. Follow these steps for precise conversions.
Step 1: Identify the Substance
The first step in converting grams to moles is identifying the chemical substance you are working with. This is crucial because each substance has a unique molar mass, which is based on its atomic or molecular composition.
For instance, if you are converting grams of sodium (Na) to moles, you must first recognize that sodium is a chemical element with the atomic symbol Na. Understanding this is the first key step to unlocking accurate conversions.
Step 2: Determine the Molar Mass
Next, you need to determine the molar mass of the substance. The molar mass is the mass of one mole of the substance and is expressed in grams per mole (g/mol). To find this value:
- Locate the atomic mass of the element on the periodic table.
- If the substance is a compound, add the atomic masses of all constituent elements.
For example, the molar mass of water (H2O) can be calculated as follows:
- Hydrogen (H) has an atomic mass of approximately 1 g/mol, and there are two hydrogen atoms.
- Oxygen (O) has an atomic mass of approximately 16 g/mol.
- Therefore, the molar mass of water = (2 * 1) + 16 = 18 g/mol.
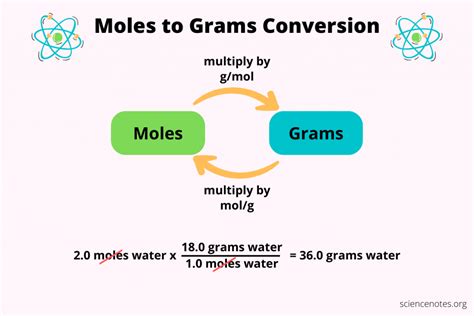
Step 3: Use the Conversion Formula
Now that you have the molar mass, you can use the formula to convert grams to moles:
moles = grams / molar mass
Let’s consider an example to clarify this step:
- Suppose you have 36 grams of water (H2O).
- Given that the molar mass of water is 18 g/mol, plug these values into the formula.
moles of water = 36 g / 18 g/mol = 2 moles
Advanced Conversion Techniques
Once you’ve mastered the basic conversion, you can explore more complex scenarios, such as dealing with mixtures and multiple substances. Here we delve into advanced methods to handle these situations.
Converting Mixtures to Moles
When dealing with mixtures, you need to convert each component to moles separately. Consider a mixture containing both sodium chloride (NaCl) and potassium chloride (KCl). First, you’ll need to convert each substance individually using the same approach as before.
- Identify the mass of each component in the mixture.
- Determine the molar mass for each substance.
- Use the conversion formula for each component.
For instance, if you have a 100-gram mixture containing 40 grams of NaCl and 60 grams of KCl:
- Molar mass of NaCl is approximately 58.44 g/mol.
- Molar mass of KCl is approximately 74.55 g/mol.
- For NaCl: moles = 40 g / 58.44 g/mol = 0.683 moles
- For KCl: moles = 60 g / 74.55 g/mol = 0.804 moles
Handling Multiple Substances in a Single Reaction
In complex chemical reactions, you may need to convert grams to moles for multiple substances. Let’s say you’re involved in a reaction where you need to determine the moles of reactants to predict product formation.
- Identify all reactants involved in the reaction.
- Calculate the moles of each reactant using the conversion formula.
- Use the stoichiometric ratios from the balanced chemical equation to find the required amounts of each substance.
For example, consider the reaction:
[ \text{N}_2 + 3 \text{H}_2 \rightarrow 2 \text{NH}_3 ]
If you have 28 grams of nitrogen (N2) and 4 grams of hydrogen (H2), convert these to moles:
- Molar mass of N2 = 28 g/mol.
- Molar mass of H2 = 2 g/mol.
- Moles of N2 = 28 g / 28 g/mol = 1 mole.
- Moles of H2 = 4 g / 2 g/mol = 2 moles.
With the stoichiometric ratios from the balanced equation, you can determine the limiting reactant and calculate the amount of product formed.
Practical FAQ
What if I make a mistake in my molar mass calculation?
Mistakes in molar mass calculations are common but easily fixable. If you realize an error, retrace your steps:
- Double-check the atomic masses of each element.
- Ensure you have correctly added or multiplied the atomic masses.
- If necessary, use a periodic table for verification.
Precision is crucial in chemistry, so take a few moments to verify your calculations to avoid discrepancies in your conversions.
How do I handle compounds with subscripts in their chemical formulas?
Compounds with subscripts, like H2O, require you to multiply the atomic mass of each element by its subscript before summing them up:
- For H2O, multiply the atomic mass of hydrogen (1 g/mol) by 2 and add it to the atomic mass of