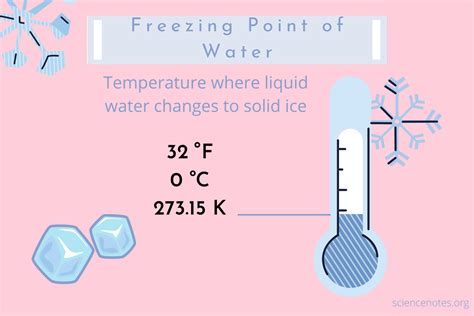
The commonly accepted freezing temperature of water has long been entrenched in scientific knowledge, but a deeper understanding reveals nuances that may surprise even seasoned professionals. It is universally taught that water freezes at 32 degrees Fahrenheit (0 degrees Celsius), but this simple notion becomes more intricate when we delve into real-world conditions. As climate change and technological advancements influence temperature readings, the foundational understanding of water’s freezing point merits re-examination.
Key Insights
- Primary insight: Under ideal laboratory conditions, water freezes at 32°F (0°C), but environmental factors significantly alter this.
- Technical consideration: The presence of impurities like salts or sugars lowers the freezing point through a phenomenon known as freezing point depression.
- Actionable recommendation: For precise measurements, consider using a calibrated, high-accuracy thermometer in a controlled environment.
Environmental Influences on Freezing Point
The freezing point of water is affected by various environmental elements that may not be present in a controlled laboratory setting. Impurities in natural water, such as salts in seawater, lower the freezing point due to a process known as freezing point depression. This concept is crucial for professionals involved in cryogenics, glaciology, or even everyday tasks like winter road maintenance. For instance, seawater typically freezes at a lower temperature than pure water, approximately 28°F (-2°C) due to dissolved salts.
Another factor is pressure. While water’s freezing point remains fairly stable at sea level, significant elevation can lead to lower freezing temperatures due to decreased atmospheric pressure. This principle is leveraged in the operation of aircraft de-icing systems, which utilize compressed air to create a pressure differential that lowers the freezing point of ice on aircraft surfaces.
The Role of Impurities and Solutes
The presence of impurities in water leads to notable changes in its freezing behavior. When substances like sugar, alcohol, or salts are mixed into water, they disrupt the orderly structure of the hydrogen-bonded network that forms ice. This disruption increases the energy required for water molecules to transition into a solid state, thereby lowering the freezing point. For instance, the addition of table salt to icy walkways creates a dilute solution that significantly lowers the freezing point, effectively melting the ice at temperatures well below 32°F (0°C).
This knowledge is not just academic; it has practical applications in fields like food preservation, where food items are packed in ice with added solutes to extend their shelf life. Additionally, understanding the effects of solutes on the freezing point of water can be crucial in medical settings, where the precise control of ice and liquid water temperatures is necessary for various treatments.
Can atmospheric conditions influence the freezing point of water?
Yes, atmospheric conditions such as pressure and the presence of impurities significantly influence the freezing point. Lower atmospheric pressure at higher altitudes can lower the freezing point, while impurities like salts create freezing point depression, lowering the temperature at which water transitions to ice.
How does freezing point depression impact road safety?
Freezing point depression is crucial in road safety, especially during winter. By adding salts to ice, municipalities lower the freezing point to ensure ice melts at lower temperatures, reducing the risk of icy roads and improving traction and safety.
Understanding the complexities surrounding the freezing temperature of water equips professionals with the knowledge needed to make informed decisions in various fields, from scientific research to everyday practical applications. By recognizing the impact of environmental factors and impurities, we gain a more nuanced comprehension of this fundamental property of water.