Mastering Ethanol Lewis Structure Explained
Understanding the Lewis structure of ethanol is crucial for grasping organic chemistry fundamentals. This guide will walk you through a detailed exploration of ethanol’s Lewis structure, with practical tips, real-world examples, and problem-solving strategies. Ethanol (C2H5OH) is a simple alcohol and serves as a foundational molecule in organic chemistry. Let’s dive into what you need to know to confidently tackle this subject.
To start with, understanding the Lewis structure of ethanol is essential not just for academic purposes but also for its practical applications in fields like pharmaceuticals, biochemistry, and environmental science. Knowing the correct Lewis structure allows chemists to predict chemical behavior, reactivity, and bonding in ethanol, making it a vital tool for professionals and students alike.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always start with the carbon atoms and connect them with single bonds to hydrogens and oxygens. This foundational structure helps ensure accuracy.
- Essential tip with step-by-step guidance: Begin by placing two carbon atoms next to each other, then add hydrogen atoms to complete their octets. Add an oxygen atom and complete its octet by adding two lone pairs and a hydrogen atom.
- Common mistake to avoid with solution: Miscounting valence electrons is a frequent error. Always double-check the total number of valence electrons before drawing the final structure.
Let’s delve into the step-by-step method for drawing the Lewis structure of ethanol, ensuring we address common pitfalls and enhance your understanding.
Step-by-Step Guide to Drawing Ethanol’s Lewis Structure
Drawing the Lewis structure for ethanol involves understanding the atomic configuration and valence electrons of each atom involved. Follow these steps to get a precise representation:
Step 1: Determine Total Valence Electrons
Each atom contributes a number of valence electrons to the molecule:
- Carbon © has 4 valence electrons each.
- Hydrogen (H) has 1 valence electron each.
- Oxygen (O) has 6 valence electrons.
To get the total number of valence electrons, sum these up:
2 Carbons x 4 = 8 electrons
6 Hydrogens x 1 = 6 electrons
1 Oxygen x 6 = 6 electrons
Total: 8 © + 6 (H) + 6 (O) = 20 valence electrons
Step 2: Form a Skeleton Structure
Place the least electronegative atom in the center. Here, carbon is less electronegative than hydrogen and oxygen, so we’ll place the two carbon atoms in the center.
Arrange the carbon atoms:
C—C
Now, attach hydrogens to the carbon atoms to satisfy the octet rule:
C—H
H—C—H
H
Attach the remaining hydrogen atoms:
H
C—H
H
H—C—H
Step 3: Attach Oxygen and Hydroxyl Group
Place the oxygen atom next to one of the carbon atoms, forming a single bond:
C—O
H
H
C—O
Attach a hydrogen atom to the oxygen:
H
C—O—H
Step 4: Distribute Remaining Electrons to Complete Octets
After placing the atoms, distribute the remaining valence electrons to complete the octets:
Start with the carbon atoms:
C—C
Each carbon now has four bonds, satisfying its octet.
Add electrons to the oxygen atom:
First, draw two lone pairs around the oxygen atom:
C—O
H
:
O:
H
Step 5: Verify the Structure
Double-check the octets:
Each carbon should have four bonds (and no lone pairs, as bonding electrons count towards the octet).
Oxygen should have eight electrons (four bonding pairs and two lone pairs).
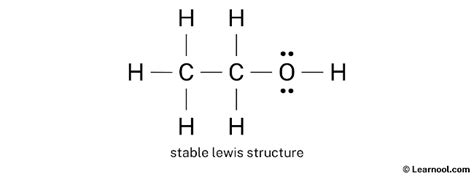
You now have a complete Lewis structure for ethanol:
H
H—C
|
H—C
O—H
|
H
Ensure you have used all 20 valence electrons.
Advanced Tips for Drawing Complex Lewis Structures
Once comfortable with ethanol’s structure, try applying these advanced techniques:
- Practice drawing other simple alcohols like methanol (CH3OH) to understand the hydroxyl group’s influence.
- Experiment with different alcohols to see how changing the alkyl group affects the structure.
- Use molecular models to visualize 3D shapes and bond angles.
Practical FAQ
What common errors should I avoid when drawing Lewis structures?
Common errors include miscounting valence electrons and forgetting to distribute them correctly. A frequent mistake is not completing the octets properly, especially for oxygen, which often requires lone pairs to satisfy the octet rule. Double-check your work and ensure all valence electrons are used.
Why is the Lewis structure important for understanding chemical properties?
The Lewis structure provides insight into the molecule’s geometry, bond types, and reactivity. For ethanol, knowing its structure helps predict its polarity, solubility, and interaction with other molecules. It is foundational for understanding more complex organic reactions.
How does the structure of ethanol differ from other alcohols?
Ethanol (C2H5OH) has a two-carbon chain with a hydroxyl group (—OH) attached to the end carbon. In other alcohols, the hydroxyl group can be attached to different positions on the carbon chain. For example, in propanol (C3H7OH), the hydroxyl group can attach to the first, second, or third carbon, leading to different isomers like 1-propanol, 2-propanol, etc. Understanding these variations helps in predicting their physical and chemical properties.
In summary, mastering the Lewis structure of ethanol not only enhances your comprehension of organic chemistry but also equips you with a practical tool to predict and understand a wide range of chemical behaviors. Keep practicing, and don’t hesitate to revisit these steps whenever you face new challenges.
This guide serves as a stepping stone for more advanced studies in chemistry, where understanding molecular structures is key to success.