When discussing the fundamental aspects of diffusion and its role in various scientific phenomena, one inevitable question arises: Does diffusion need energy? To fully address this, we must delve into the core principles of diffusion, explore its mechanisms, and understand how it operates in different contexts. This guide aims to provide clear, actionable insights and practical examples, guiding you through the intricacies of diffusion and how it functions without requiring energy input.
Diffusion is a spontaneous process that occurs in any system where molecules or particles are in motion. It is driven by the natural tendency to achieve uniformity in concentration, driven by thermal energy. Let's explore the core aspects of diffusion by addressing the main concerns users often have, providing clear solutions, practical examples, and easy-to-follow instructions to enhance your understanding of this crucial process.
Problem-Solution Opening Addressing User Needs
Have you ever wondered why certain things happen naturally over time, like the mixing of different gases in a room or the slow spread of a scent through the air? These occurrences are all examples of diffusion at work, but they often leave us curious about why they happen and how. The misconception that diffusion requires an energy input is quite common; it's essential to understand that diffusion itself does not consume energy but rather relies on the inherent kinetic energy of particles to spread out evenly across a medium. This guide aims to demystify the process by providing a clear, practical understanding of diffusion, complete with actionable insights and real-world examples to ensure you can easily apply this knowledge to your own scenarios.
Quick Reference
- Immediate action item with clear benefit: Check the thermal conditions around any system you observe. Since diffusion is driven by kinetic energy, understanding the temperature will give you insights into the diffusion rate.
- Essential tip with step-by-step guidance: Use Fick's Laws of Diffusion to calculate the rate of diffusion in various mediums. Start with Fick’s first law for a simple gradient-based diffusion process.
- Common mistake to avoid with solution: Don’t assume that diffusion always leads to faster mixing. Factors like concentration gradient and medium viscosity play a crucial role; optimizing these can enhance diffusion without energy input.
Understanding Diffusion: Principles and Mechanisms
To grasp diffusion at a fundamental level, we must delve into the principles that govern it. Diffusion occurs due to the random motion of particles driven by thermal energy. This motion causes particles to spread out from areas of high concentration to areas of low concentration until equilibrium is reached. Here's a detailed breakdown of how diffusion works and why it doesn't require an energy input from external sources.
The Fundamental Nature of Diffusion
Diffusion is the passive movement of particles (such as ions, atoms, or molecules) from a region of higher concentration to a region of lower concentration due to their kinetic energy. This process continues until the concentration is uniform throughout the medium. There are several types of diffusion, each with specific applications and implications:
- Simple Diffusion: Movement of molecules through a concentration gradient without any energy input.
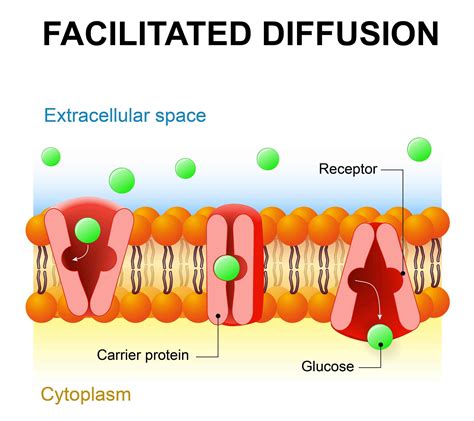
- Facilitated Diffusion: Involves carrier proteins or channels to help molecules cross membranes, but still driven by concentration gradient.
- Osmosis: A type of diffusion specific to water molecules moving across a selectively permeable membrane.
- Diffusion in Gases: Particles move freely in gases due to low density and high kinetic energy.
Diffusion does not require energy because it's a natural, spontaneous process driven by the kinetic energy of particles. The inherent thermal motion of molecules means they constantly collide and move in random directions. When there’s a concentration gradient, molecules naturally move from higher to lower concentration regions until they’re evenly distributed.
Why Diffusion Doesn’t Need Energy
To understand why diffusion does not require energy input, consider the second law of thermodynamics, which states that systems tend to move from a state of higher entropy to a state of lower entropy without external work. Diffusion aligns with this principle by moving particles from areas of higher concentration (higher energy) to areas of lower concentration (lower energy), thus reducing local entropy until equilibrium is achieved.
Moreover, kinetic energy in a system comes from the temperature. Higher temperatures increase the kinetic energy of particles, accelerating the diffusion process. This intrinsic energy within the particles means no additional energy input is necessary for diffusion to occur.
How Diffusion Works: A Practical Guide
Understanding the operational mechanisms of diffusion can be crucial in various fields such as chemistry, biology, and physics. Here's a step-by-step breakdown that includes practical examples to demonstrate the process and principles of diffusion:
Step-by-Step Mechanism of Diffusion
1. Identify the Particles: Determine the particles involved. For example, in biological systems, this might be oxygen diffusing into cells.
2. Check Concentration Gradient: Assess whether there is a concentration gradient. A gradient exists when particles are more concentrated in one area than another.
3. Observe Movement: Track how particles move. They naturally move from higher concentration areas to lower concentration areas due to kinetic energy.
4. Monitor Over Time: Observe the process over time to see how the concentration evens out.
Example: Consider the diffusion of ink in water. When a drop of ink is added to water, it begins to spread out over time until it's uniformly mixed with the water. This occurs because the ink molecules move randomly due to thermal energy until they are evenly distributed.
Practical Examples of Diffusion
Here are a few examples to further elucidate the principles of diffusion:
- In the Atmosphere: When a person exhales into the air, the carbon dioxide from their breath slowly diffuses through the air until it’s uniformly mixed with the surrounding air.
- In Biological Systems: Oxygen from the lungs diffuses into the bloodstream and then to body tissues until it is evenly distributed.
- In Laboratory Experiments: When potassium permanganate is added to water, it rapidly diffuses until it is uniformly distributed, showcasing the efficiency of diffusion in liquids.
Best Practices for Enhancing Diffusion
To optimize the diffusion process in various applications, consider these best practices:
- Increase Temperature: Higher temperatures increase kinetic energy, speeding up diffusion.
- Reduce Concentration Gradient: Try to minimize the initial concentration difference to accelerate the diffusion process.
- Choose Suitable Mediums: Some mediums facilitate faster diffusion. For instance, gases diffuse faster than liquids due to lower particle density.
- Use Porous Materials: Materials with small pores can enhance the rate of diffusion by providing pathways for particles to move.
By following these guidelines, you can better understand and harness the principles of diffusion without requiring any external energy input.
Practical FAQ: Addressing User Pain Points
Can Diffusion Occur in a Vacuum?
Yes, diffusion can occur in a vacuum. While it's slower due to the lack of particles to interact with, gas molecules will still diffuse from areas of high concentration to low concentration until they are evenly distributed. This principle can be observed in processes like the outgassing of materials over time.
How Does Pressure Affect Diffusion?
Pressure affects diffusion, especially in gases. Higher pressure can increase the concentration of particles in a given volume, which in turn can accelerate diffusion if the pressure difference is significant. In practical terms, gases will diffuse more rapidly in a higher pressure environment compared to a lower pressure one.
Is Diffusion Always Slow?
No, diffusion isn’t always slow. The rate of diffusion is influenced by several factors including the temperature, particle size, concentration gradient,