Carbon’s crystalline form, diamond, not only captivates with its dazzling brilliance but also holds profound importance in both scientific and industrial realms. Understanding the diamond chemical formula is paramount for those in geology, materials science, and various industrial applications. This article dives deep into the essential aspects of diamonds, offering expert insights and practical applications.
Key Insights
- Diamonds are composed of a single element, carbon, arranged in a crystalline structure.
- Understanding the sp3 hybridization in diamonds is crucial for applications in electronics.
- Synthetic diamonds are increasingly used in cutting tools and high-performance applications.
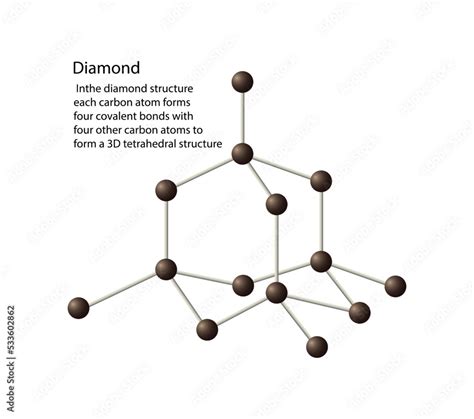
Structure of Diamond Chemical Formula
Diamonds are fundamentally a form of carbon, with the chemical formula C. Under immense pressure and high temperatures, carbon atoms arrange themselves into a tetrahedral lattice. This crystalline structure is the foundation of a diamond’s notable properties such as exceptional hardness and high thermal conductivity. The sp3 hybridization of carbon atoms is critical here: each carbon atom bonds with four others, creating a robust, three-dimensional network. This molecular architecture is responsible for diamonds’ unparalleled strength and durability. For example, this unique structure allows diamonds to serve as the hardest naturally occurring material, making them ideal for cutting and grinding tools.Applications of the Diamond Chemical Formula
In the industrial sector, diamonds are leveraged extensively due to their distinctive properties derived from the diamond chemical formula. High precision in manufacturing is enhanced by diamond-tipped tools, which are pivotal in industries like aerospace and electronics. In the realm of electronics, synthetic diamonds doped with boron create p-type semiconductors, playing a role in advancing electronic devices. This application underscores the importance of understanding the diamond’s structure at a molecular level to innovate in technology and industry. The ability to manipulate and understand the diamond chemical formula has enabled the development of cutting tools, abrasives, and even in the creation of high-performance semiconductor materials.Are natural and synthetic diamonds different?
Natural diamonds and synthetic diamonds differ primarily in their formation process rather than their chemical formula. Both are composed of carbon atoms in a crystalline structure, but synthetic diamonds are created under controlled conditions in laboratories, whereas natural diamonds form over millions of years deep within the Earth's mantle.
How is the diamond chemical formula relevant in industry?
The diamond chemical formula is crucial for industrial applications due to its inherent properties. The unique molecular structure allows diamonds to be used in cutting tools, abrasives, and as a substrate for high-performance electronic devices. Understanding the diamond chemical formula is essential for optimizing these industrial uses and for the development of synthetic diamonds.
Exploring the diamond chemical formula reveals its immense scientific and industrial significance. It’s the fundamental understanding of carbon’s arrangement that drives the application of diamonds in advanced technology and high-precision tools. As we continue to delve deeper into the molecular intricacies, the potential applications and innovations promise to further revolutionize multiple sectors. In conclusion, diamonds stand as a testament to the power of nature’s chemistry and the advancements achievable through scientific comprehension.