Are you struggling to accurately measure the density of ethanol for your scientific, educational, or industrial needs? You’re not alone. Ethanol density measurement can be tricky due to various factors like temperature, pressure, and ethanol’s intrinsic properties. But fear not! This comprehensive guide will provide you with step-by-step, practical advice to achieve precise ethanol density measurements. Let’s dive in and unravel the secrets of precise ethanol density measurement.
Introduction: Why Accurate Ethanol Density Measurement Matters
Accurate measurement of ethanol density is critical across a range of applications—from environmental science to chemical engineering. Incorrect measurements can lead to errors in formulation, inefficient industrial processes, and potentially even safety hazards. By mastering the art of precise ethanol density measurement, you’ll ensure your processes run smoothly, your data is reliable, and your results are accurate.Quick Reference
Quick Reference
- Immediate action item: Always calibrate your equipment before starting your measurements to ensure accuracy.
- Essential tip: Use a digital hydrometer designed for alcohol solutions to get precise readings.
- Common mistake to avoid: Failing to account for temperature variations when measuring ethanol density.
Step-by-Step Guidance for Measuring Ethanol Density
To achieve precise ethanol density measurements, it’s important to follow a series of detailed steps. Below, we break down the process into manageable sections for clarity and ease of implementation.Section 1: Preparation
Start by preparing all necessary tools and materials before beginning your measurement:
- Digital hydrometer
- Thermometer
- Ethanol sample
- Graduated cylinder or volumetric flask
- Calibrated scale (optional, for mass-based density)
Ensure all equipment is clean and free from contaminants. Contamination can significantly affect your measurements.
Section 2: Calibration
Before taking any measurements, calibrate your hydrometer. This ensures the device is functioning correctly:
Place the hydrometer in distilled water at a known temperature (usually 20°C or 68°F). The hydrometer should float at a specific density value corresponding to pure water. Adjust your device if needed.
Section 3: Sample Preparation
Properly prepare your ethanol sample:
- Fill a clean graduated cylinder or volumetric flask to the mark with your ethanol sample.
- Make sure the sample is well mixed to avoid localized concentration differences.
Record the volume of your ethanol sample accurately for later use.
Section 4: Temperature Control
Temperature plays a crucial role in ethanol density measurements. Follow these steps:
Using a thermometer, measure the temperature of your ethanol sample in degrees Celsius (°C). Record this temperature precisely. Ethanol density changes with temperature, so accurate temperature measurement is essential.
Section 5: Measurement
Now you’re ready to take the density measurement:
- Place the hydrometer in the ethanol sample carefully. Make sure it’s fully submerged without touching the bottom of the container.
- Wait a few moments for the hydrometer to float levelly. Observe the mark on the hydrometer that aligns with the surface of the ethanol.
- Record this density reading. Remember to convert the reading according to the temperature correction if needed.
If using mass to calculate density, you will also need to measure the mass of the ethanol sample with a calibrated scale.
Section 6: Correction and Calculation
To get the most accurate density, correct for temperature and calculate as necessary:
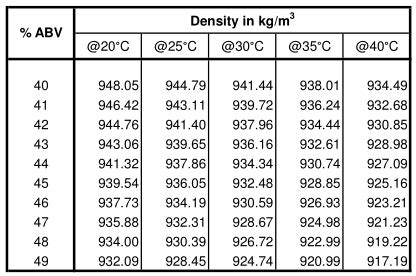
- Use a temperature correction chart or formula to adjust your initial density reading. This accounts for the effect of temperature on ethanol density.
- If mass was used, calculate density using the formula:
- Density (ρ) = Mass (m) / Volume (V)
For ethanol, typically the formula is applied as:
- Density (g/mL) = Mass (grams) / Volume (mL)
Practical FAQ
What should I do if my hydrometer sinks too much?
If your hydrometer sinks excessively, it might be that the ethanol sample is not mixed well. Re-dissolve the ethanol thoroughly. Additionally, ensure there’s no residual contamination on the hydrometer or container.
Can I use a regular hydrometer for ethanol?
Regular hydrometers aren’t typically designed for ethanol solutions. It’s best to use a hydrometer specifically calibrated for alcohol solutions to ensure precise readings.
How do I account for temperature variations?
Temperature correction for ethanol density is critical. Use a temperature correction chart specific to ethanol or an online calculator that factors in the temperature of your ethanol sample to adjust your measurements accordingly.
By following these detailed instructions, you’ll be well-equipped to measure ethanol density accurately. This guide offers a step-by-step approach starting from basic preparations to more advanced corrections and calculations. Stay tuned for continued improvements and keep your measurements precise and reliable.