Cofactor vs Coenzyme: Unveiling Biological Mysteries
Understanding the intricate dance of biochemical reactions that occur within living organisms can often feel overwhelming. This guide dives deep into the world of biological molecules, specifically focusing on cofactors and coenzymes—two critical players in the grand scheme of biochemical processes. Whether you’re a student, a professional scientist, or simply someone fascinated by the inner workings of life at a molecular level, this guide will provide you with clear, actionable advice and practical solutions.
Understanding Cofactors and Coenzymes: The Essentials
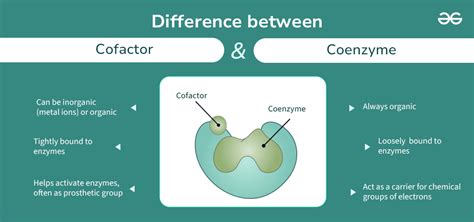
To get started, it’s crucial to differentiate between cofactors and coenzymes, as they are essential for enzyme activity but serve distinct roles in biochemical reactions.
Cofactors are non-protein molecules that assist enzymes in performing their functions. They can be either inorganic ions or organic compounds. In contrast, coenzymes are organic compounds that act as cofactors. While both cofactors and coenzymes are vital to enzyme function, coenzymes typically have a more prominent role in catalytic activities due to their involvement in redox reactions.
Why It Matters
Understanding cofactors and coenzymes is not just an academic exercise—it has real-world applications in medicine, pharmacology, and even agriculture. For instance, deficiencies in certain vitamins that serve as coenzymes can lead to diseases like scurvy (Vitamin C deficiency) or beriberi (Vitamin B1 deficiency). Moreover, knowledge of these molecules is fundamental in developing drugs that target specific enzymes, potentially leading to treatments for diseases ranging from cancer to metabolic disorders.
Quick Reference
Quick Reference
- Immediate action item: Identify if a molecule is a cofactor or coenzyme by checking if it’s an organic compound (coenzyme) or inorganic (cofactor).
- Essential tip: Remember that coenzymes often bind reversibly to enzymes, playing crucial roles in redox reactions.
- Common mistake to avoid: Confusing cofactors and coenzymes. Always check if the molecule is organic (coenzyme) or inorganic (cofactor).
Cofactors: The Inorganic Assistants
Inorganic cofactors are mineral ions that are integral to enzyme function. Here, we will explore their types, roles, and examples to better understand how they operate in biological systems.
Types of Inorganic Cofactors
Inorganic cofactors can be metal ions such as magnesium (Mg2+), iron (Fe2+, Fe3+), and zinc (Zn2+).
Role of Inorganic Cofactors
These ions play a crucial role in stabilizing enzyme structure, facilitating redox reactions, and aiding in the binding of substrates to the enzyme's active site. For example, magnesium ions are essential in the enzyme RuBisCO, which is pivotal in the photosynthesis process.
Examples of Inorganic Cofactors
Let's delve into a few examples to provide clarity:
- Magnesium (Mg2+): Magnesium ions are crucial in DNA replication, acting as a cofactor for DNA polymerase, which synthesizes new strands of DNA.
- Zinc (Zn2+): Zinc ions are important in various enzymes such as carbonic anhydrase, which catalyzes the reversible hydration of carbon dioxide, and alcohol dehydrogenase, involved in alcohol metabolism.
How to Identify Inorganic Cofactors
To determine if an inorganic molecule is an inorganic cofactor, follow these steps:
- Check if the molecule is an ion.
- Determine if it binds to an enzyme, thereby facilitating its catalytic activity.
- Verify if it participates in redox reactions or stabilizes the enzyme's structure.
Coenzymes: The Organic Helpers
Coenzymes are organic molecules that assist enzymes in catalyzing biochemical reactions. Unlike cofactors, they are not part of the enzyme's structure but bind to the enzyme temporarily to enhance its catalytic activity.
Types of Coenzymes
Coenzymes can be classified into two main categories: those that participate in redox reactions and those that act as carriers of chemical groups.
- Redox Coenzymes: These include NAD+, FAD, and Coenzyme A, which are involved in the transfer of electrons or hydrogen atoms.
- Adenylate Coenzymes: These carry chemical groups such as amino groups or phosphate groups. Examples include ATP and certain nucleotide coenzymes.
Role of Coenzymes
Coenzymes play a vital role in various metabolic pathways. They often serve as electron carriers, energy carriers, or group carriers. For instance, NAD+ and FAD facilitate the transfer of electrons in cellular respiration, a crucial process for energy production.
Examples of Coenzymes
Here are some notable coenzymes and their functions:
- NAD+: Nicotinamide adenine dinucleotide is involved in redox reactions in glycolysis, the citric acid cycle, and oxidative phosphorylation.
- FAD: Flavin adenine dinucleotide is essential in the citric acid cycle and various other redox reactions.
- Coenzyme A: It carries acyl groups in various metabolic pathways, including fatty acid oxidation.
How to Identify Coenzymes
To identify coenzymes, consider the following steps:
- Determine if the molecule is organic.
- Confirm if it temporarily binds to an enzyme.
- Check if it participates in redox reactions or carries chemical groups.
Practical Applications of Cofactors and Coenzymes
In real-world scenarios, understanding cofactors and coenzymes can lead to practical applications in fields like medicine, pharmacology, and biotechnology. Here’s how you can apply this knowledge:
Medical Applications
Identifying coenzyme deficiencies can lead to diagnostic tests and treatment plans for vitamin-related diseases. For instance, monitoring levels of coenzyme Q10 in patients with mitochondrial disorders can guide therapeutic strategies.
Pharmacological Applications
Drugs that target specific enzymes can be developed to treat diseases. For example, statins target HMG-CoA reductase, an enzyme in the cholesterol synthesis pathway, thereby reducing cholesterol levels.
Biotechnological Applications
Enzymes and their cofactors can be harnessed in industrial processes to produce bioproducts like biofuels, bioplastics, and pharmaceuticals. Understanding the cofactor requirements for specific enzymes can optimize these bioprocesses.
Practical FAQ
How do cofactors and coenzymes differ?
Cofactors are non-protein molecules that assist enzymes in their function. They can be inorganic ions or organic compounds. Coenzymes are a type of organic cofactor that is essential in catalytic activities, especially redox reactions. Cofactors are broadly categorized based on whether they are inorganic (metal ions) or organic (coenzymes). While all coenzymes are cofactors, not all cofactors are coenzymes.
What is an example of a cofactor in enzyme activity?
An example of a cofactor in enzyme activity is the magnesium ion (Mg2+). Magnesium ions often play a critical role in stabilizing enzyme structures and in facilitating the binding of substrates to the enzyme’s active site. For instance, in the enzyme DNA polymerase, Mg2+ ions help in the replication process by stabilizing the enzyme and the DNA strand.