Unlocking the Secrets of ClF3 Lewis Structure: A Detailed Guide
To delve into the intricacies of ClF3, one must understand the fundamental principles governing its Lewis structure. Chlorine trifluoride (ClF3) is a fascinating molecule that combines elements of both theoretical chemistry and practical application. As an expert in molecular structure and bonding, I will unravel the detailed insights required to comprehend this molecule. The complexity of ClF3’s Lewis structure is not just academic; it has significant implications for fields like molecular chemistry and industrial applications.
Key Insights
- ClF3 features an irregular T-shaped geometry due to the presence of lone pair electrons on the central chlorine atom.
- To depict its Lewis structure correctly, it’s crucial to consider the octet rule and the ability of chlorine to expand its octet.
- Understanding the hybridization and electron distribution is essential for practical applications in fields like molecular modeling and chemical synthesis.
Understanding the Lewis Structure of ClF3
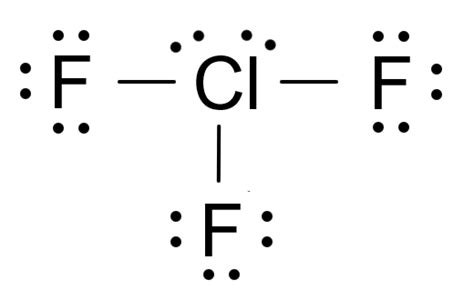
The Lewis structure of ClF3 begins with the central chlorine atom. This atom forms three covalent bonds with three fluorine atoms, utilizing three of its valence electrons. However, chlorine has five valence electrons, and three of them are used for bonding, leaving two unbonded electrons, or lone pairs, which are critical for determining its molecular shape. To accommodate these lone pairs and form an expanded octet, chlorine adopts sp3d hybridization, a concept that allows it to exceed the traditional octet rule. These lone pairs repel the bonding pairs, leading to the molecule’s unique T-shaped geometry, which is less symmetrical than other chlorine compounds due to the influence of the lone pairs.
Hybridization and Electron Geometry
Understanding the hybridization in ClF3 is crucial for grasping its electron geometry. Chlorine, in ClF3, undergoes sp3d hybridization, which is necessary to account for the five regions of electron density (three bonding pairs and two lone pairs). This hybridization allows the chlorine atom to form three bonds with fluorine and accommodate the lone pairs in a way that minimizes electron-electron repulsion. The presence of these lone pairs also means that the molecular geometry differs from the electron pair geometry. While the electron pair geometry is trigonal bipyramidal, the molecular geometry, as dictated by the positioning of the lone pairs, is T-shaped.
Applications and Implications
ClF3 is not just an intriguing molecule for academic purposes; it has significant real-world applications. One notable use is in nuclear power, where ClF3 is employed in the purification of uranium and other nuclear fuels due to its ability to form volatile fluoride compounds with actinides. This property makes ClF3 invaluable in nuclear fuel reprocessing and waste treatment. Moreover, understanding the Lewis structure and molecular geometry helps chemists predict the reactivity and stability of ClF3, which is pivotal for its safe handling and application in industrial processes.
Why does ClF3 have a T-shaped geometry?
ClF3 has a T-shaped geometry due to the presence of two lone pairs on the central chlorine atom, which repel the three bonding pairs, causing a distortion from the expected trigonal bipyramidal electron pair geometry.
Can ClF3 form bonds beyond the traditional octet rule?
Yes, ClF3 can form an expanded octet as chlorine utilizes sp3d hybridization, allowing it to bond with three fluorine atoms while accommodating two lone pairs on the central atom.
Understanding ClF3’s Lewis structure and its associated geometry and hybridization offers significant insights into both theoretical and practical chemistry, ensuring safe and effective use in various industrial applications.