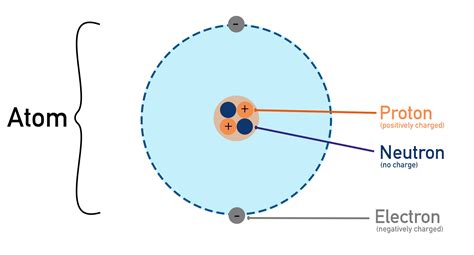
Charged atoms play a crucial role in modern physics and chemistry, influencing everything from electronic devices to chemical reactions. An atom becomes charged when it gains or loses electrons, creating ions with a net positive or negative charge. This fundamental concept underpins various technological innovations and scientific breakthroughs. Understanding charged atoms allows us to decipher phenomena such as electricity, magnetism, and the behavior of materials at the atomic level. This article delves into the science of charged atoms, exploring their characteristics, applications, and implications in practical scenarios.
The Nature of Charged Atoms
Charged atoms, or ions, arise when an atom gains or loses electrons, resulting in a net charge. This charge can be either positive, due to a loss of electrons, or negative, due to an acquisition of electrons. The stability and reactivity of these ions are pivotal for a multitude of chemical reactions. For example, sodium chloride, common table salt, is formed through the ionic bond between the positively charged sodium ion (Na+) and the negatively charged chloride ion (Cl-). This interaction not only defines the compound’s properties but also impacts its use in biological systems, such as maintaining fluid balance in the body.
Applications of Charged Atoms in Technology
The practical applications of charged atoms span several advanced technological fields. One significant application is in the semiconductor industry, where the controlled doping of silicon with charged atoms, such as phosphorus or boron, allows for the creation of diodes and transistors—foundational components of modern electronics. Another fascinating application is in medical imaging, where charged atoms and ions are utilized in techniques such as MRI (Magnetic Resonance Imaging). Charged particles in the human body interact with magnetic fields, producing signals that are converted into detailed images of internal structures.
Key Insights
Key Insights
- Primary insight with practical relevance: Charged atoms influence a wide range of scientific and technological fields.
- Technical consideration with clear application: Understanding ion formation and properties enhances the development of advanced materials.
- Actionable recommendation: Invest in advanced research to explore the applications of charged atoms in new technological innovations.
Chemical Reactions Driven by Charged Atoms
Chemical reactions are often driven by the interactions of charged atoms. When two ions with opposite charges come into proximity, they form ionic bonds, which are fundamental to many compounds we use daily. For instance, the acidic properties of vinegar, primarily acetic acid (CH3COOH), are a result of the charged hydrogen ions (H+) it donates during a chemical reaction. Additionally, the formation of water (H2O) involves the combination of charged hydrogen ions (H+) and oxygen atoms (O). The charge distribution and stability of these ions make them essential in determining the outcome of various chemical processes.
FAQ Section
What role do charged atoms play in electrical conductivity?
Charged atoms are critical for electrical conductivity as they allow for the movement of electrons, which is the basis of electric current. Metals, which contain free electrons, exhibit high electrical conductivity due to the mobility of these charged particles.
How do charged atoms affect material properties?
Charged atoms influence material properties significantly. For example, the doping of silicon with charged atoms can change its electrical properties, allowing it to function as a semiconductor. This alteration is fundamental in the design of electronic components.
Understanding the science behind charged atoms opens the door to innovative technological advancements and deeper scientific insights. Their role in chemical reactions, technological applications, and material properties is indispensable, underscoring their critical importance in both scientific research and practical applications.