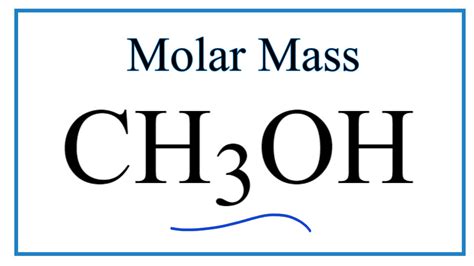
Understanding the exact molar mass of methanol, or CH3OH, is a fundamental concept in chemistry. This seemingly simple calculation is pivotal for many chemical reactions, from organic synthesis to determining the concentration of solutions in biochemistry. This guide will walk you through each step of finding the molar mass of methanol and highlight its crucial role in chemical processes.
If you’re a chemist, student, or even a chemical enthusiast, you’ve likely encountered methanol in labs and literature. Whether you're measuring reagent amounts, developing new compounds, or analyzing solutions, knowing the exact molar mass of CH3OH (methanol) is essential. This guide will break down the calculation, offer practical applications, and address common challenges.
Why Knowing the Molar Mass Matters
The molar mass of a compound is the weight of one mole of that substance. For methanol, CH3OH, it’s a key piece of information that aids in stoichiometry, a vital part of chemical equations and reactions. Understanding its molar mass allows chemists to precisely measure reactants, predict product amounts, and design experiments efficiently. Let’s dive into why this calculation is so important and how you can do it accurately.
For example, in organic synthesis, the molar mass enables you to translate grams of reactants into moles, ensuring that you use the correct proportions for successful reactions. In biochemistries, it helps in determining how much of a particular reagent to add to a solution to achieve a desired concentration.
Quick Reference
Quick Reference
- Immediate action item: Calculate the molar mass of CH3OH by summing the atomic masses of its constituent elements.
- Essential tip: Use a periodic table to find the atomic masses of carbon, hydrogen, and oxygen.
- Common mistake to avoid: Confusion between molecular formula and empirical formula; ensure you understand the complete molecular formula.
How to Calculate the Molar Mass of CH3OH
Let’s get into the nuts and bolts of calculating the molar mass. Here’s a step-by-step guide that even beginners can follow to ensure precision:
- Identify the molecular formula of CH3OH: Methanol has the molecular formula CH3OH, consisting of one carbon (C) atom, three hydrogen (H) atoms, and one oxygen (O) atom.
- Find atomic masses: Look up the atomic masses of each element from a reliable periodic table:
- Carbon (C): 12.01 g/mol
- Hydrogen (H): 1.01 g/mol per atom
- Oxygen (O): 16.00 g/mol
- Calculate the molar mass:
Add the atomic masses together:
- Carbon: 1 x 12.01 g/mol = 12.01 g/mol
- Hydrogen: 4 x 1.01 g/mol = 4.04 g/mol
- Oxygen: 1 x 16.00 g/mol = 16.00 g/mol
Sum these values to get the total molar mass:
12.01 g/mol (C) + 4.04 g/mol (H) + 16.00 g/mol (O) = 32.05 g/mol
Therefore, the exact molar mass of CH3OH is 32.05 g/mol.
Real-world Applications of CH3OH Molar Mass
Now that we’ve covered the basics, let’s delve into the practical applications where knowing the molar mass of methanol plays a critical role:
Organic Synthesis
In organic chemistry, methanol is a widely used solvent and reagent. For instance, when synthesizing esters, you’ll need to accurately weigh methanol to ensure the right concentration for the reaction.
Biochemistry
In biochemical assays, methanol is often part of the buffer solutions. Knowing its molar mass allows precise preparation of these solutions, essential for maintaining the integrity of experimental setups.
Pharmaceutical Development
Methanol is crucial in the development of various pharmaceutical compounds. The accurate molar mass aids in creating formulations and ensuring proper dosage levels in drugs.
Detailed How-To Sections
Calculating Molar Mass in Various Compounds
The process for calculating molar mass is universal, not just for methanol, but for any compound. Here’s a detailed how-to guide that applies to any chemical formula:
- Step 1: Write the chemical formula: Begin with the precise chemical formula of the compound. For methanol, it’s CH3OH.
- Step 2: Use a periodic table: Refer to a periodic table to find the atomic masses of each element present in the formula.
- Step 3: Multiply the atomic mass by the number of atoms: For each element, multiply its atomic mass by the number of atoms it has in the compound.
- Step 4: Sum the total mass: Add up all the individual totals to get the molar mass of the entire compound.
Example Calculations
Let’s consider some examples to solidify this concept:
Example 1: Water (H2O)
- Hydrogen: 2 x 1.01 g/mol = 2.02 g/mol
- Oxygen: 1 x 16.00 g/mol = 16.00 g/mol
- Total: 2.02 g/mol + 16.00 g/mol = 18.02 g/mol
Example 2: Sodium chloride (NaCl)
- Sodium: 1 x 22.99 g/mol = 22.99 g/mol
- Chlorine: 1 x 35.45 g/mol = 35.45 g/mol
- Total: 22.99 g/mol + 35.45 g/mol = 58.44 g/mol
Advanced Considerations
For more complex molecules, consider additional factors:
- Isotopes: Natural abundance of isotopes might affect molar mass, especially in large scale or precise applications.
- Hydration states: Some compounds come with water of hydration. Ensure to account for these when calculating molar mass.
Practical FAQ
Why is it important to know the molar mass?
Knowing the molar mass is essential for several reasons:
- Stoichiometry: It allows for accurate measurements and calculations in chemical reactions.
- Concentration: Precise molar mass calculation helps in determining the concentration of solutions.
- Scaling reactions: Accurate molar mass values help in scaling up reactions from lab to industrial scale.
For instance, if you need to prepare a solution with a specific concentration, knowing the molar mass ensures that the quantity you measure out will achieve the desired concentration.
What common mistakes should I avoid when calculating molar mass?
Here are some common pitfalls and their solutions: