Understanding the molar mass of chemicals is fundamental for anyone delving into the realm of chemistry. Among various compounds, C3H8 or propane, stands out due to its significance in both natural and industrial settings. This article will guide you through the precise calculation of C3H8 molar mass, incorporating practical insights and real examples to enhance your grasp of this essential concept.
The Fundamental Concept of Molar Mass
Molar mass is the mass of a given substance (chemical element or chemical compound) divided by the amount of substance (in moles). This measurement is pivotal in chemical reactions and stoichiometry, enabling chemists to predict the amount of reactants needed or products formed. The molar mass of a compound is essentially the sum of the atomic masses of all the atoms present in the molecular formula.Breaking Down C3H8
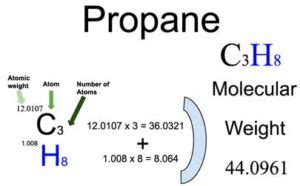
Propane, represented as C3H8, consists of three carbon © atoms and eight hydrogen (H) atoms. To determine its molar mass, one must refer to the periodic table and utilize the atomic masses: carbon © has an atomic mass of approximately 12.01 g/mol, while hydrogen (H) has an atomic mass of about 1.008 g/mol.Key Insights
- Primary insight with practical relevance: Understanding the molar mass calculation of C3H8 is critical for balancing chemical equations and scaling up reactions in a lab setting.
- Technical consideration with clear application: Knowing the molar mass allows chemists to convert between grams and moles, crucial for determining the yield and efficiency of chemical processes.
- Actionable recommendation: Always double-check your calculations to avoid common errors, ensuring accuracy in your chemical computations.
Step-by-Step Calculation
To accurately calculate the molar mass of propane (C3H8), follow these steps:- Identify the atomic masses of the constituent elements. For propane, carbon (C) weighs 12.01 g/mol and hydrogen (H) weighs 1.008 g/mol.
- Multiply the atomic mass of each element by the number of atoms of that element in the compound. Propane has 3 carbon atoms and 8 hydrogen atoms.
- Sum the total masses. The total molar mass is calculated as follows: (3 atoms of carbon × 12.01 g/mol) + (8 atoms of hydrogen × 1.008 g/mol) = 36.03 g/mol + 8.064 g/mol = 44.094 g/mol.
Real-World Application
Understanding C3H8’s molar mass is not just an academic exercise; it has practical implications in various fields. For example, in the industrial production of chemicals, knowing the molar mass is vital for determining the precise amount of reactants needed to produce a desired amount of product. Additionally, propane is used as a fuel in various applications, from domestic heating to powering vehicles. Accurate calculations ensure efficient fuel usage and environmental safety.Why is the molar mass of a compound important?
The molar mass of a compound is crucial because it allows for the conversion between mass and moles, which is essential for balancing chemical equations, determining reactant and product quantities, and ensuring efficient use of materials in chemical processes.
How can mistakes in calculating molar mass affect experimental results?
Mistakes in molar mass calculations can lead to incorrect quantities of reactants being used, which may drastically alter the outcome of a chemical reaction. This can affect the yield, purity of the product, and even the safety of the experimental setup.
In conclusion, calculating the molar mass of C3H8 or any compound requires meticulous attention to detail. By understanding and applying this fundamental concept, chemists can enhance their precision and efficiency in both laboratory and industrial settings.