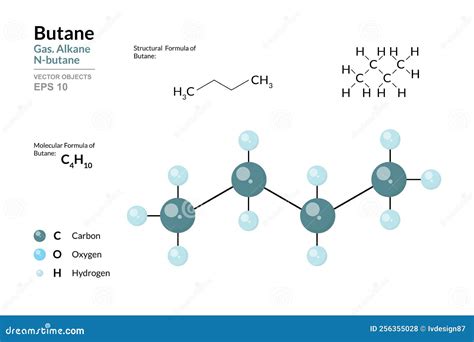
The molecular formula for butane, C₄H₁₀, represents a fundamental component in organic chemistry and industrial chemistry, primarily known as a primary constituent of liquefied petroleum gas (LPG). This simple yet significant hydrocarbon compound’s structure and properties play a critical role in various applications, ranging from fuel to organic synthesis. Understanding butane’s molecular formula and structural insights can provide valuable insights into its behavior and applications in different fields.
Key insights box:
Key Insights
- Butane’s molecular formula is C₄H₁₀, highlighting its composition as a four-carbon alkane.
- The compound exhibits structural isomerism, featuring both straight-chain and branched forms.
- As a gas at standard conditions, butane serves as an efficient and clean fuel, underpinning its use in portable stoves and gas grills.
Understanding butane’s molecular formula necessitates a grasp of its structure and properties. Butane is a linear alkane with a molecular weight of 58.12 g/mol. The formula indicates a molecule composed of four carbon atoms (C₄) bonded to ten hydrogen atoms (H₁₀). The carbon atoms form a straight chain, which can be either in a n-butane (normal butane) configuration or as isobutane (2-methylpropane). The distinction lies in the position of a methyl group (-CH₃) attached to the second carbon atom in isobutane, creating a branched structure. This structural difference profoundly impacts butane’s physical properties, such as boiling point and reactivity.
The significance of butane’s molecular formula extends beyond its simple representation. The compound is a prime example of alkane isomerism, showcasing the importance of structural configuration in determining physical and chemical behavior. The n-butane form has a boiling point of -0.5°C, making it a suitable gas at standard atmospheric conditions. Isobutane, on the other hand, has a slightly higher boiling point of -11.7°C. This variation is crucial for industrial applications, especially in cooling and refrigeration systems, where the phase state of butane is critical.
Butane’s molecular formula also emphasizes its role as a vital energy source. When combusted, butane releases energy efficiently due to its high hydrogen-to-carbon ratio. This makes it a preferred fuel in portable applications. The combustion reaction for butane is straightforward:
2C₄H₁₀ + 13O₂ → 8CO₂ + 10H₂O
This exothermic reaction underscores the utility of butane as a reliable fuel source in scenarios where portability and efficiency are paramount.
FAQ section
What are the different forms of butane?
Butane exists in two primary structural forms: n-butane and isobutane. n-Butane is a straight-chain alkane, while isobutane is a branched structure with a methyl group attached to the second carbon atom.
Why is butane used as a fuel?
Butane is used as a fuel due to its high energy release during combustion, high hydrogen-to-carbon ratio, and liquid-to-gas phase transition at standard conditions, making it ideal for portable stoves and grills.
In conclusion, the butane molecular formula encapsulates more than just a set of numbers and letters. It unveils the chemical’s diverse applications, from fuel to synthetic chemistry, highlighting its importance in both industrial and everyday contexts. The knowledge of butane’s structure and properties equips professionals and enthusiasts alike with the understanding needed to harness its potential effectively and safely.