Cellular energy is the fundamental currency of life, underpinning all cellular functions. Two critical molecules at the heart of this energy system are adenosine triphosphate (ATP) and adenosine diphosphate (ADP). Understanding the interplay between ATP and ADP is essential for anyone delving into cellular biology, biochemistry, or physiology. This article delves into the differences, functions, and importance of ATP and ADP in cellular energy management.
The Role of ATP in Cellular Energy
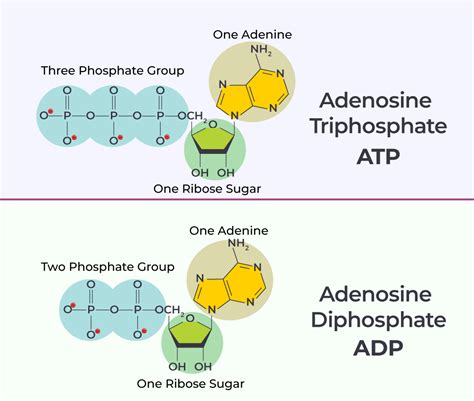
Adenosine triphosphate (ATP) is often hailed as the powerhouse of the cell, a key player in energy transactions. ATP is composed of adenosine and three phosphate groups, enabling it to store high-energy phosphate bonds. When the terminal phosphate bond is hydrolyzed, it releases a substantial amount of energy, which the cell can use for various activities such as muscle contraction, nerve impulse propagation, and biosynthesis.
ATP as the Primary Energy Currency
ATP’s primary insight with practical relevance lies in its role as the cell’s direct energy currency. It provides the necessary energy for active transport mechanisms, protein synthesis, and cellular division. For example, the conversion of glucose through cellular respiration produces ATP, which powers muscle contraction during physical activity.
Key Insights
- ATP functions as the direct energy currency of the cell.
- Technically, ATP releases energy upon the hydrolysis of its high-energy phosphate bonds.
- Actionable recommendation: Focus on cellular respiration pathways to understand ATP production.
The Function of ADP in Energy Dynamics
Adenosine diphosphate (ADP) is the product left behind when ATP loses one phosphate group. While ATP grabs the spotlight for its high-energy role, ADP is essential in the regeneration of ATP. ADP combines with a phosphate group, typically through the energy released from the cell’s metabolic processes, to reform ATP, thus continuing the cycle.
ADP in the ATP-ADP Cycle
ADP is an indispensable component of the ATP-ADP cycle. Understanding ADP’s technical consideration with clear application involves recognizing its role in the regeneration phase of the cycle. When ATP donates energy to a cellular process, it becomes ADP plus an inorganic phosphate (Pi), a reaction that powers cellular functions. Conversely, ADP can be reactivated by capturing a phosphate group from substrate-level phosphorylation or oxidative phosphorylation, reforming ATP and sustaining energy supply.
FAQ Section
Is ATP always present in high concentrations?
No, ATP levels in cells are tightly regulated and fluctuate according to the cell's energy needs. High levels of ATP indicate active energy production, while low levels suggest increased energy demand.
How do cells regenerate ATP?
Cells regenerate ATP through two main pathways: substrate-level phosphorylation and oxidative phosphorylation. These processes involve the capture of inorganic phosphate to ADP, converting it back to ATP.
In the realm of cellular biochemistry, ATP and ADP form an intricate energy dynamic essential for life. The primary insight here is that while ATP serves as the immediate energy provider, ADP’s role in the ATP-ADP cycle is equally critical for maintaining this energy equilibrium. This understanding underscores the importance of both molecules in cellular processes and highlights practical steps for anyone studying the biochemical underpinnings of cellular function.