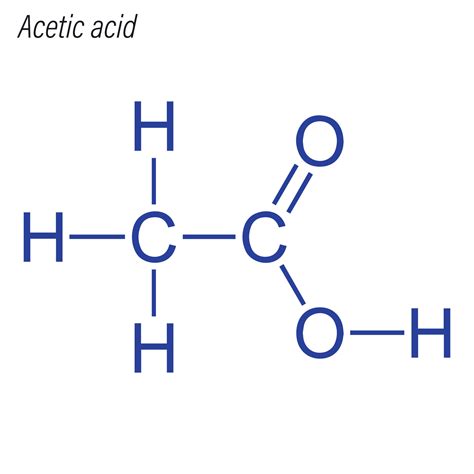
The acetic acid molecule, denoted as CH₃COOH in chemical nomenclature, plays an essential role in both organic chemistry and industrial applications. Understanding the Lewis structure of acetic acid provides valuable insights into its chemical behavior, reactivity, and interactions. This guide aims to uncover the intricacies of acetic acid’s Lewis structure through a lens of practical insights, evidence-based statements, and real-world examples.
Key Insights
- Acetic acid consists of a methyl group (CH₃) and a carboxyl group (COOH), both of which are crucial for understanding its Lewis structure.
- The carboxyl group’s oxygen atoms are pivotal for hydrogen bonding and other significant chemical interactions.
- Drawing the accurate Lewis structure aids in predicting the molecule's molecular geometry and understanding its dipole moment.
Structure of Acetic Acid
Acetic acid’s Lewis structure is fundamental to grasping its chemistry. The molecule is composed of a four-carbon chain with one of the carbons bearing a methyl group (CH₃) and the other having a carboxyl group (COOH). The structure starts with carbon-carbon single bonds linking the atoms together. The methyl group comprises one carbon atom bonded to three hydrogen atoms. The carboxyl group involves a carbon atom doubly bonded to an oxygen atom and singly bonded to another oxygen atom, with the latter also bonded to a hydrogen atom.This arrangement ensures that the carbon atoms are tetrahedrally bonded, fulfilling the octet rule. The oxygen in the carboxyl group has two lone pairs, and the hydrogen in the OH group has none. These electron arrangements highlight the molecule’s dual functionality, allowing it to act both as a weak acid and a hydrogen bond acceptor.
Role of the Carboxyl Group
The carboxyl group in acetic acid, COOH, is central to its Lewis structure and functionality. Oxygen’s high electronegativity dictates its behavior in the molecule. One of the oxygen atoms forms a double bond with the carbon atom, while the other forms a single bond and possesses a hydroxyl group (OH). The double-bonded oxygen atom has two lone pairs of electrons, whereas the hydroxyl oxygen has one lone pair.The presence of these lone pairs on the oxygen atoms means the molecule can engage in hydrogen bonding, a significant aspect in various physical properties like boiling point and solubility. Hydrogen bonding is facilitated by the electronegativity difference between hydrogen and oxygen, resulting in a partial positive charge on hydrogen and a partial negative charge on oxygen.
Analysis and Practical Implications
Analyzing the Lewis structure of acetic acid reveals its dual nature as a molecule: it is both an acid and a hydrogen bond donor. The carboxyl group’s ability to donate a proton (H⁺) in solutions aligns with its acidic properties. This acidity makes acetic acid useful in various applications, such as in vinegar, which is about 5-8% acetic acid in water.Moreover, the Lewis structure provides an insight into the molecule’s dipole moment. The uneven distribution of electrons around the central carbon atoms leads to a net dipole, which is a critical factor in determining the molecule’s polarity and its interaction with polar solvents.
What is the significance of the carboxyl group in acetic acid?
The carboxyl group in acetic acid is significant because it not only imparts acidic properties to the molecule, enabling it to donate protons in solutions but also facilitates hydrogen bonding. This group is crucial for understanding the molecule's chemical reactivity and physical properties such as boiling point and solubility.
How does the Lewis structure help predict acetic acid's behavior?
The Lewis structure of acetic acid aids in predicting its molecular geometry, polarity, and its ability to participate in hydrogen bonding. Understanding the distribution of electrons and lone pairs helps in predicting how the molecule will interact with other molecules in various chemical reactions and solutions.
In conclusion, the detailed examination of the Lewis structure of acetic acid unveils a comprehensive understanding of its chemical and physical properties. The insights derived from this structure are invaluable for both academic and industrial applications.